Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
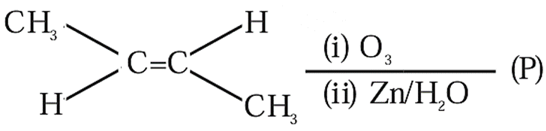
Consider the given reaction. Trans-2-butene $$\xrightarrow{(i) O_3}$$ $$\xrightarrow{(ii) Zn/H_2O}$$ (P)
The total number of oxygen atoms present per molecule of the product (P) is _______.
Correct Answer: 1
The reagents $$O_3$$ followed by $$Zn/H_2O$$ indicate reductive ozonolysis. In this reaction, the carbon-carbon double bond is cleaved completely, and the resulting fragments are converted into aldehydes or ketones.
The given alkene is trans-2-butene, $$CH_3-CH=CH-CH_3$$. Cleavage of the $$C=C$$ bond divides the molecule into two identical fragments. Each fragment forms acetaldehyde, $$CH_3CHO$$. Therefore, the product obtained is acetaldehyde.
A single molecule of acetaldehyde contains one oxygen atom.
Hence, the number of oxygen atoms present in one molecule of product $$P$$ is $$1$$
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
