Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Two flasks I and II shown below are connected by a valve of negligible volume.
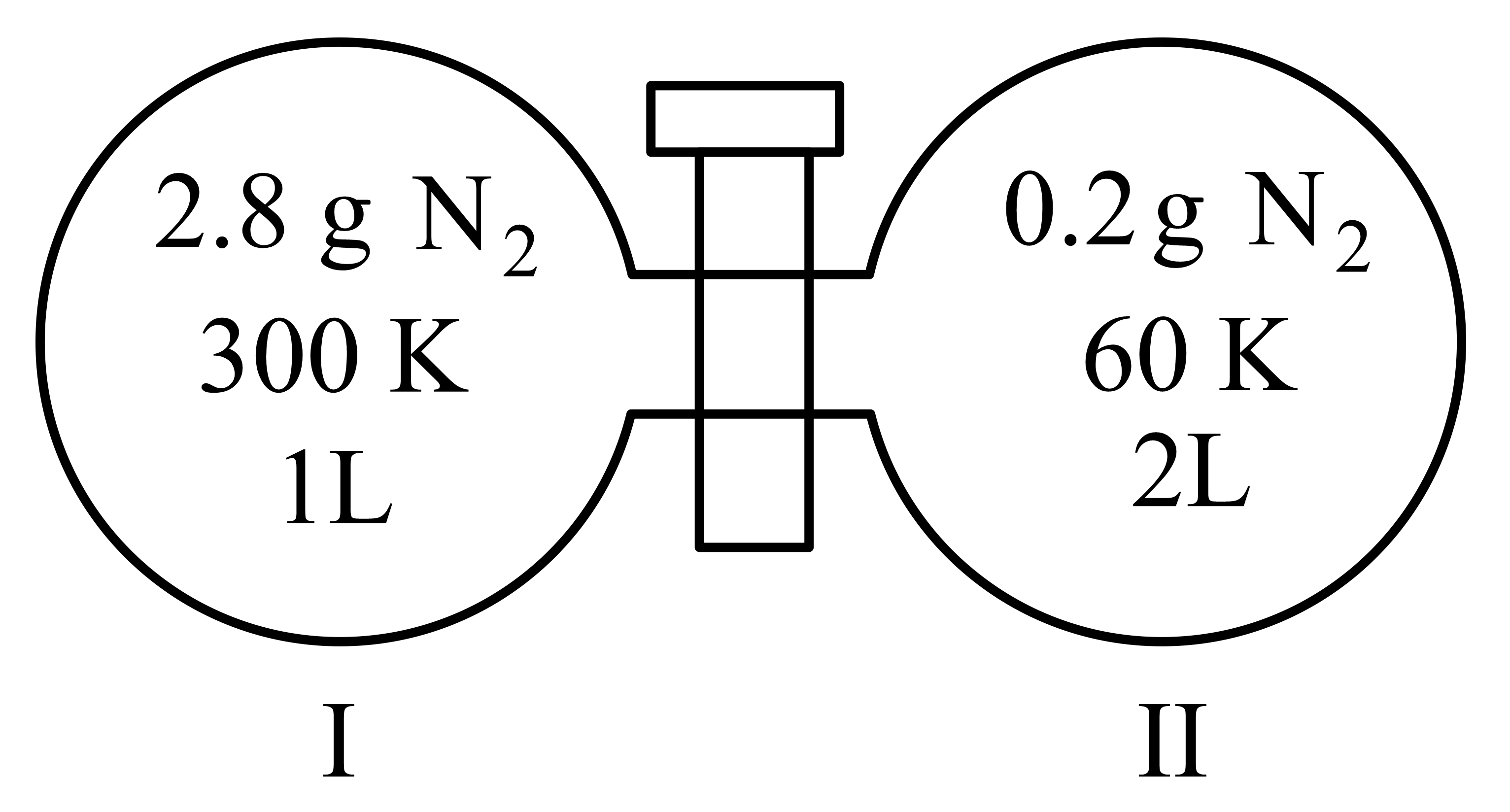
When the valve is opened, the final pressure of the system in bar is $$x \times 10^{-2}$$. The value of x is _________. (Integer answer)
[Assume : Ideal gas; 1 bar = $$10^5$$ Pa; Molar mass of N$$_2$$ = 28.0 mol$$^{-1}$$; R = 8.31 J mol$$^{-1}$$ K$$^{-1}$$]
Correct Answer: 84
$$n_1 = \frac{2.8}{28} = 0.1\ mol$$
$$n_2 = \frac{0.2}{28} = 0.00714\ mol$$
Since the system is insulated,
$$n_1T_1 + n_2T_2 = (n_1+n_2)T_f$$
$$T_f = \frac{n_1T_1+n_2T_2}{n_1+n_2}$$
$$T_f = \frac{(0.1)(300)+(0.00714)(60)}{0.1+0.00714}$$
$$T_f = \frac{30+0.4284}{0.10714}$$
$$T_f = 284K$$
$$V_f = 1L + 2L = 3L = 3\times10^{-3}m^3$$
$$n_f = \frac{2.8}{28}+\frac{0.2}{28} = 0.10714\ mol$$
$$P_f = \frac{n_fRT_f}{V_f}$$
$$P_f = \frac{(0.10714)(8.31)(284)}{3\times10^{-3}}$$
$$P_f = 8.41\times10^4Pa$$
$$1bar = 10^5Pa$$
$$P_f = \frac{8.41\times10^4}{10^5}$$
$$P_f = 0.841bar$$
$$P_f = 84\times10^{-2}bar$$
$$\therefore x = 84$$
Create a FREE account and get:
Educational materials for JEE preparation
