Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the above reaction. The percentage yield of amide product is ________ (Round off to the Nearest Integer).
(Given: Atomic mass: C: 12.0u, H: 1.0u, N: 14.0u, O: 16.0u, Cl: 35.5u)
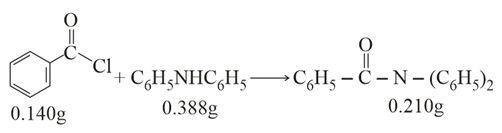
Correct Answer: 77
To determine the percentage yield of the amide product, we first calculate the molar masses of the compounds involved using the given atomic masses, which are
$$140.5 \text{ g/mol}$$for benzoyl chloride ($$C_7H_5OCl$$), $$169 \text{ g/mol}$$ for diphenylamine ($$C_{12}H_{11}N$$), and $$273 \text{ g/mol}$$
for the resulting amide product ($$C_{19}H_{15}NO$$). Next, we find the number of moles for both reactants to identify the limiting reagent. The moles of benzoyl chloride are calculated as
$$\frac{0.140 \text{ g}}{140.5 \text{ g/mol}} \approx 0.000996 \text{ mol}$$, while the moles of diphenylamine are $$\frac{0.388 \text{ g}}{169 \text{ g/mol}} \approx 0.002296 \text{ mol}$$. Since $$0.001 < 0.002296$$, benzoyl chloride is the limiting reagent and dictates the maximum amount of product that can be formed. Using the 1:1 stoichiometric ratio, the theoretical yield of the amide product is $$0.001 \text{ mol} \times 273 \text{ g/mol} \approx 0.272 \text{ g}$$.
Finally, we calculate the percentage yield by dividing the actual given yield ($$0.210 \text{ g}$$) by the theoretical yield ($$0.272 \text{ g}$$) and multiplying by 100, which gives$$\left( \frac{0.210}{0.272} \right) \times 100 \approx 76.9\%$$. Rounding off to the nearest integer, the final percentage yield is 77.
Create a FREE account and get:
Educational materials for JEE preparation
