Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
If one mole of an ideal gas at $$(P_1, V_1)$$ is allowed to expand reversibly and isothermally (A to B) its pressure is reduced to one-half of the original pressure (see figure). This is followed by a constant volume cooling till its pressure is reduced to one-fourth of the initial value $$(B \to C)$$. Then it is restored to its initial state by a reversible adiabatic compression (C to A). The net work done by the gas is equal to:
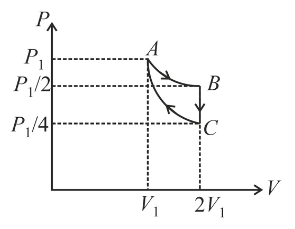
For the complete cycle,
$$W_{\text{net}}=W_{AB}+W_{BC}+W_{CA}$$
For A→B, expansion is isothermal.
For one mole,
$$W_{AB}=RT\ln\frac{V_B}{V_A}$$Since at A,
$$RT=P_1V_1$$and
$$V_B=2V_1$$
so
$$W_{AB}=P_1V_1\ln2$$
For B→CB \to CB→C, volume is constant, so
$$W_{BC}=0$$
For C→A, process is adiabatic compression.
For adiabatic process,
Q=0
and
$$W=\frac{P_iV_i-P_fV_f}{\gamma-1}$$For compression from C to A,
$$W_{CA}=\frac{P_CV_C-P_AV_A}{\gamma-1}$$
Now
$$P_CV_C=\frac{P_1}{4}(2V_1)=\frac{P_1V_1}{2}$$and
$$P_AV_A=P_1V_1$$
Thus
$$W_{CA}=\frac{\frac{P_1V_1}{2}-P_1V_1}{\gamma-1}$$$$=-\frac{P_1V_1}{2(\gamma-1)}$$So net work done is
$$W_{\text{net}}=P_1V_1\ln2-\frac{P_1V_1}{2(\gamma-1)}$$$$W_{\text{net}}=RT\ln2-\frac{P_1V_1}{2(\gamma-1)}$$
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.