Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
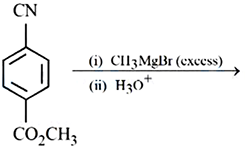
Major product of the following reaction is: A compound with $$CO_2CH_3$$ group reacts with (i) $$CH_3MgBr$$ (excess) (ii) $$H_3O^+$$
In excess CH3-MgBr, firstly the ester group gets reduced to substituted alcohol then the Grignard's reagent attacks on the CN group first converting it to imine and then upon addition of water the result is formation of ketone. Since, the aforementioned imine salt is stable it does not undergo further Grignard reagent attacks and upon hydrolysis Oxygen being more electronegative takes up the double bond resulting in removal of -NH2 group in form of Ammonia.
Create a FREE account and get:
Educational materials for JEE preparation
