Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
For a double strand DNA, one strand is given below:
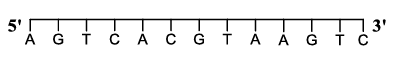
The amount of energy required to split the double strand DNA into two single strands is ______ kcal $$mol^{-1}$$.
[Given: Average energy per H-bond for A-T base pair = 1.0 kcal $$mol^{-1}$$, G-C base pair = 1.5 kcal $$mol^{-1}$$, and A-U base pair = 1.25 kcal $$mol^{-1}$$. Ignore electrostatic repulsion between the phosphate groups.]
Correct Answer: 41
The given strand of the double-stranded DNA is
$$5' \; A \; G \; T \; C \; A \; C \; G \; T \; A \; A \; G \; T \; C \; 3'$$
Step 1 : Count the number of each nucleotide in this strand.
A (adenine) = 4 (at positions 1, 5, 9, 10)
T (thymine) = 3 (at positions 3, 8, 12)
G (guanine) = 3 (at positions 2, 7, 11)
C (cytosine) = 3 (at positions 4, 6, 13)
Step 2 : Identify the number of base pairs.
An A on one strand pairs with T on the complementary strand, and vice-versa; this gives an A-T base pair wherever the original strand has A or T. Similarly, G pairs with C, giving a G-C base pair wherever the original strand has G or C.
Number of A-T base pairs $$= 4 + 3 = 7$$
Number of G-C base pairs $$= 3 + 3 = 6$$
Step 3 : Convert “energy per H-bond” to “energy per base pair”.
Each A-T pair contains 2 H-bonds, energy per bond $$= 1.0\; \text{kcal}\, \text{mol}^{-1}$$
Energy per A-T pair $$= 2 \times 1.0 = 2.0\; \text{kcal}\, \text{mol}^{-1}$$
Each G-C pair contains 3 H-bonds, energy per bond $$= 1.5\; \text{kcal}\, \text{mol}^{-1}$$
Energy per G-C pair $$= 3 \times 1.5 = 4.5\; \text{kcal}\, \text{mol}^{-1}$$
Step 4 : Calculate the total energy required to break all H-bonds in the double strand.
Total energy $$= (7 \times 2.0) + (6 \times 4.5)$$
$$= 14.0 + 27.0$$
$$= 41.0\; \text{kcal}\, \text{mol}^{-1}$$
Hence, the energy required to separate the two DNA strands is 41 kcal mol−1.
Create a FREE account and get:
Educational materials for JEE preparation
