Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
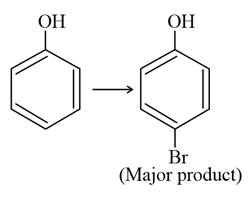
The given reaction can occur in the presence of:
(a) Bromine water
(b) $$Br_{2}$$ in $$CS_{2}$$, 273 K
(c) $$Br_{2} / FeBr_{3}$$
(d) $$Br_{2}$$ in $$CHCl_{3}$$, 273 K
Choose the correct answer from the options given below:
Halogenation: On treating phenol with bromine, different reaction products are formed under different experimental conditions.
(a) When the reaction is carried out in solvents of low polarity such as $$\mathrm{CHCl_3}$$ or $$\mathrm{CS_2}$$ and at low temperature, monobromophenols are formed.
$$\mathrm{C_6H_5OH + Br_2 \xrightarrow[\ 273\ K\ ]{CS_2} o\text{-}Bromophenol + p\text{-}Bromophenol}$$
The usual halogenation of benzene takes place in the presence of a Lewis acid such as $$\mathrm{FeBr_3}$$, which polarises the halogen molecule. In case of phenol, the polarisation of bromine molecule takes place even in the absence of Lewis acid due to the highly activating effect of the $$\mathrm{-OH}$$ group attached to the benzene ring.
(b) When phenol is treated with bromine water, $$\mathrm{2,4,6}$$-tribromophenol is formed as a white precipitate.
$$\mathrm{C_6H_5OH + 3Br_2 \rightarrow C_6H_2Br_3OH + 3HBr}$$
$$\mathrm{2,4,6\text{-}Tribromophenol}$$
Thus b, c, d give the right options. a gives a different product. Thus, the right option is C.

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
