Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
For a certain metal, when monochromatic light of wavelength $$\lambda$$ is incident, the stopping potential for photoelectrons is $$3V_0$$. When the same metal is illuminated by light of wavelength $$2\lambda$$, then the stopping potential becomes $$V_0$$. The threshold wavelength for photoelectric emission for the given metal is $$\alpha \lambda$$. The value of $$\alpha$$ is __________.
For photoemission, Einstein’s equation is
$$\frac{hc}{\lambda}\;=\;\phi\;+\;eV_s$$
where $$\phi$$ is the work-function of the metal and $$V_s$$ is the stopping potential.
Case 1: Incident wavelength $$\lambda$$ gives stopping potential $$3V_0$$.
$$\frac{hc}{\lambda}\;=\;\phi\;+\;e(3V_0)$$ $$-(1)$$
Case 2: Incident wavelength $$2\lambda$$ gives stopping potential $$V_0$$.
$$\frac{hc}{2\lambda}\;=\;\phi\;+\;e(V_0)$$ $$-(2)$$
Subtract $$(2)$$ from $$(1)$$:
$$\frac{hc}{\lambda}-\frac{hc}{2\lambda}\;=\;e(3V_0)-e(V_0)$$
Left side: $$\frac{hc}{\lambda}\left(1-\frac12\right)=\frac{hc}{2\lambda}$$
Right side: $$2eV_0$$
Hence
$$\frac{hc}{2\lambda}=2eV_0\quad\Longrightarrow\quad hc = 4eV_0\lambda$$ $$-(3)$$
Insert $$hc$$ from $$(3)$$ into equation $$(2)$$ to find the work-function:
$$\frac{4eV_0\lambda}{2\lambda}=\phi+eV_0$$
$$2eV_0=\phi+eV_0$$
$$\phi = eV_0$$ $$-(4)$$
The threshold (cut-off) wavelength $$\lambda_0$$ is defined by $$\frac{hc}{\lambda_0}=\phi$$. Using $$(3)$$ and $$(4)$$:
$$\frac{4eV_0\lambda}{\lambda_0}=eV_0$$ $$\frac{4\lambda}{\lambda_0}=1$$ $$\lambda_0 = 4\lambda$$
Thus $$\alpha = 4$$.
Option B which is: $$4$$
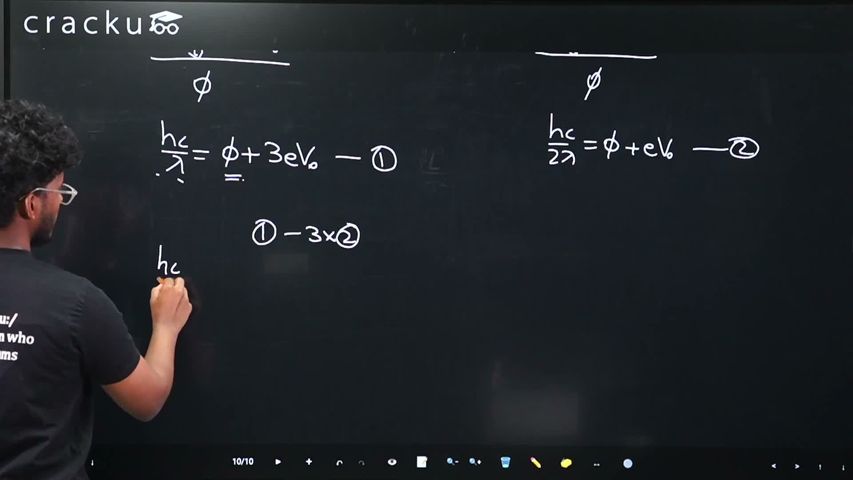
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
