Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
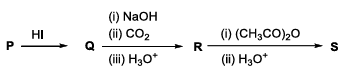
In the given reaction scheme, P is a phenyl alkyl ether, Q is an aromatic compound; R and S are the major products.
The correct statement about S is
When one side of an ether is aryl and the other side is alkyl, hot concentrated $$HI$$ selectively cleaves the alkyl-oxygen bond (because the aryl-oxygen bond has partial double-bond character). Therefore:
$$\text{Phenyl-O-R} \xrightarrow{HI} \text{Phenol }(Q)\;+\;RI$$
Thus $$Q$$ is phenol, $$C_6H_5OH$$.
The Kolbe-Schmitt reaction is the standard industrial route for converting phenol into salicylic acid. The three-step sequence is:
$$C_6H_5OH\; \xrightarrow[\text{(ii) }CO_2]{\text{(i) NaOH, 140-160 °C, 4-7 atm}} \xrightarrow[\;]{\text{(iii) }H_3O^+} o\text{-hydroxybenzoic acid}$$
Hence $$R$$ is salicylic acid, commonly written as $$o\text{-}HO-C_6H_4-COOH$$.
Acetic anhydride acetylates the phenolic -OH of salicylic acid much faster than the -COOH group. The two-step sequence
$$o\text{-}HO-C_6H_4-COOH \xrightarrow[\text{(ii) }H_3O^+]{\text{(i) }(CH_3CO)_2O} o\text{-}AcO-C_6H_4-COOH$$
gives $$S$$ = acetylsalicylic acid, better known as aspirin.
Pharmacologically, aspirin irreversibly acetylates and inactivates cyclo-oxygenase (COX-1 and COX-2). These enzymes catalyse the conversion of arachidonic acid into prostaglandins. By blocking this step, aspirin inhibits the biosynthesis of prostaglandins, which are mediators of pain, fever and inflammation.
Therefore, the correct statement about $$S$$ is:
Option B which is: It inhibits the synthesis of prostaglandin.
Create a FREE account and get:
Educational materials for JEE preparation
