Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The correct order for acidity of the following hydroxyl compound is
(A) CH$$_3$$OH
(B) (CH$$_3$$)$$_3$$COH
(C)
(D)
(E)
Choose the correct answer from the options given below:
We need to arrange the given hydroxyl compounds in order of decreasing acidity.
The acidity of a hydroxyl compound ($$R-OH$$) depends on the stability of the conjugate base ($$R-O^-$$) formed after loss of the proton. The more stable the conjugate base, the stronger the acid.
(A) $$CH_3OH$$ - Methanol (aliphatic alcohol)
(B) $$(CH_3)_3COH$$ - tert-Butanol (aliphatic alcohol)
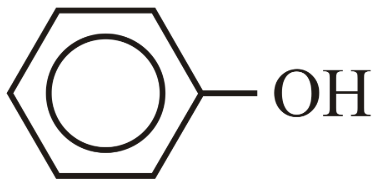
(C) Phenol ($$C_6H_5OH$$)
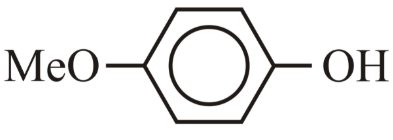
(D) p-MeO-Phenol ($$4-CH_3O-C_6H_4-OH$$)
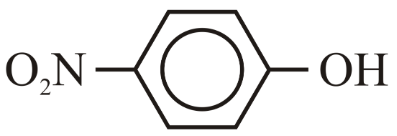
(E) p-$$NO_2$$-Phenol ($$4-NO_2-C_6H_4-OH$$)
Phenols are significantly more acidic than aliphatic alcohols because the phenoxide ion ($$C_6H_5O^-$$) is stabilized by resonance with the aromatic ring. The negative charge is delocalized over the ring. Aliphatic alkoxide ions have no such resonance stabilization. Therefore: All phenols > All alcohols in acidity.
Order among the phenols (E, C, D).
- p-$$NO_2$$-Phenol (E): The $$NO_2$$ group is a strong electron-withdrawing group (both by inductive and resonance effects). It stabilizes the phenoxide ion by pulling electron density away from the oxygen, delocalizing the negative charge further. This makes it the most acidic phenol.
- Phenol (C): The unsubstituted phenol, with moderate acidity.
- p-MeO-Phenol (D): The $$OCH_3$$ group is an electron-donating group by resonance (lone pairs on oxygen donate into the ring). This destabilizes the phenoxide ion by increasing electron density, making it less acidic than unsubstituted phenol. Therefore: E > C > D.
Order among the alcohols (A, B).
- $$CH_3OH$$ (A): Methanol has one methyl group, which is weakly electron-donating by hyperconjugation/induction.
- $$(CH_3)_3COH$$ (B): tert-Butanol has three methyl groups donating electron density to the carbon bearing the $$OH$$. This destabilizes the alkoxide ion more than in methanol. Therefore: A > B in acidity.
$$E > C > D > A > B$$p-$$NO_2$$-Phenol > Phenol > p-MeO-Phenol > $$CH_3OH$$ > $$(CH_3)_3COH$$
The correct answer is Option 4: E > C > D > A > B.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.