Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The decreasing order of hydride affinity for following carbocations is:
(a)
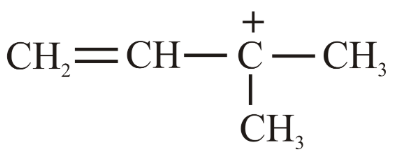
(b)
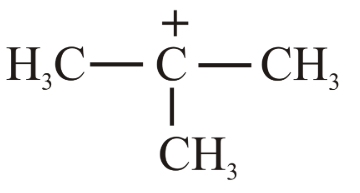
(c)
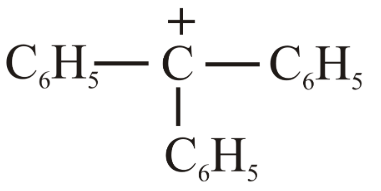
(d)
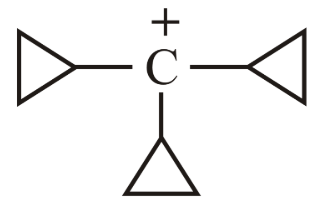
Choose the correct answer from the options given below:
Hydride affinity is defined for the reaction
$$R^+ + H^- \;\rightarrow\; RH$$.
The larger the energy released (that is, the more negative the reaction enthalpy), the greater is the hydride affinity of the carbocation $$R^+$$.
A carbocation that is intrinsically less stable gains a larger amount of stabilisation on accepting a hydride ion, so it shows a higher hydride affinity.
Conversely, a more stable carbocation is already low in energy; it gains relatively little additional stabilisation on accepting $$H^-$$ and therefore exhibits a lower hydride affinity.
Hence,
$$\text{higher hydride affinity}\; \Longleftrightarrow\; \text{lower inherent stability of the carbocation}$$.
Case 1 (c):The methyl cation $$CH_3^+$$ possesses neither hyperconjugation nor resonance stabilisation. It is the least stabilised of the four; therefore it will have the highest hydride affinity.
Case 2 (a):The n-propyl (1°) carbocation $$CH_3CH_2CH_2^+$$ is stabilised by hyperconjugation with two α-hydrogen atoms. It is more stable than $$CH_3^+$$ but less stable than a 3° or resonance-stabilised cation. Its hydride affinity is therefore lower than that of (c) but higher than that of (b) and (d).
Case 3 (b):The tert-butyl (3°) carbocation $$(CH_3)_3C^+$$ enjoys extensive hyperconjugation (nine α-hydrogen atoms), making it markedly more stable than a 1° carbocation. Hence its hydride affinity is lower than those of (c) and (a).
Case 4 (d):The benzyl carbocation $$C_6H_5CH_2^+$$ is stabilised by resonance with the aromatic ring, distributing the positive charge over several atoms. This resonance stabilisation is stronger than the hyperconjugative stabilisation present in $$(CH_3)_3C^+$$. Therefore it is the most stable of the four and consequently has the lowest hydride affinity.
Combining the four cases, the order from highest to lowest hydride affinity is
$$\text{(c)} \; \gt \; \text{(a)} \; \gt \; \text{(b)} \; \gt \; \text{(d)}.$$
This corresponds to Option B: C, A, B, D.
Answer : Option B
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.