Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Here is the information broken down into well-formatted paragraphs to make it much easier to read and study:
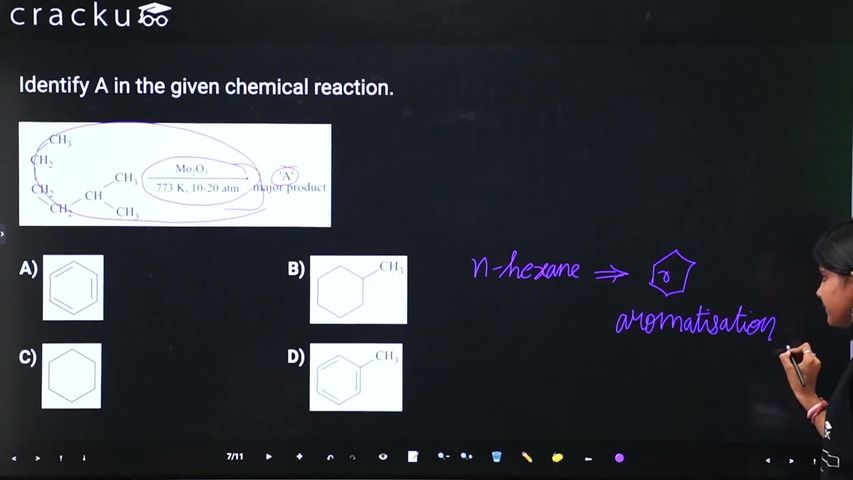
When n-alkanes containing six or more carbon atoms are subjected to high temperatures of 773 K and pressures of 10 to 20 atmospheres, they undergo a major structural transformation. This process requires the presence of specific catalysts, typically oxides of vanadium, molybdenum, or chromium supported over an alumina base.
Under these intense conditions, the straight-chain molecules undergo simultaneous dehydrogenation (the loss of hydrogen atoms) and cyclization (the forming of a ring). This converts the alkanes into benzene and its homologues. In organic chemistry, this transformative reaction is formally known as aromatization or catalytic reforming.
To illustrate this with a specific example, if the goal is to synthesize toluene (which is a seven-carbon homologue of benzene), a seven-carbon alkane such as 2-methylhexane or normal heptane can be used as the starting reactant.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
