Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
In the following p- V diagram the equation of state along the curved path is given by $$(V-2)^{2}=4ap$$ where a is a constant. The total work done in the closed path is
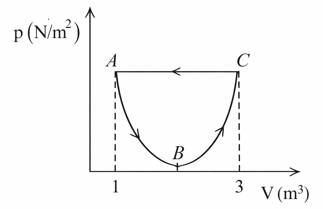
Net work done in a cyclic process equals area enclosed by the loop.
Upper path C→A is horizontal (constant pressure), and lower path is given by
$$(V-2)^2=4ap$$
Write pressure as
$$p=\frac{(V-2)^2}{4a}$$
At points A and C,
$$V=1,V=3$$
Substitute into curve:
For V=1,
$$p=\frac{(1-2)^2}{4a}=\frac{1}{4a}$$
For V=3,
$$p=\frac{(3-2)^2}{4a}=\frac{1}{4a}$$
So upper straight line has pressure
$$p=\frac{1}{4a}$$
Now work done over cycle equals area between straight line and curve:
$$W=\int_1^3\left(\frac{1}{4a}-\frac{(V-2)^2}{4a}\right)dV$$
Take $$\frac{1}{4a}$$ common:
$$W=\frac{1}{4a}\int_1^3\left(1-(V-2)^2\right)dV$$
Put
x=V−2
Then limits become
V=1→x=−1
V=3→x=1
So
$$W=\frac{1}{4a}\int_{-1}^1(1-x^2)dx$$
Integrating,
$$=\frac{1}{4a}\left[x-\frac{x^3}{3}\right]_{-1}^1$$
$$=\frac{1}{4a}\left(\frac{2}{3}-\left(-\frac{2}{3}\right)\right)$$
$$=\frac{1}{4a}\cdot\frac{4}{3}$$
$$=\frac{1}{3a}$$
The arrows show process is
A→B→C→A
During compression, pressure is higher than during expansion, so work done on the gas is greater than work done by the gas.
Therefore net work done by the gas is negative.
So,
W=−(enclosed area)
$$W=-\frac{1}{3a}$$
Create a FREE account and get:
Educational materials for JEE preparation
