Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
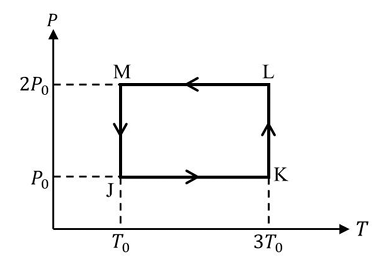
One mole of a monatomic ideal gas undergoes the cyclic process J $$\to$$ K $$\to$$ L $$\to$$ M $$\to$$ J, as shown in the P-T diagram.
Match the quantities mentioned in List-I with their values in List-II and choose the correct option.
[$$R$$ is the gas constant.]
| List-I | List-II | ||
|---|---|---|---|
| (P) | Work done in the complete cyclic process | (1) | $$RT_0 - 4RT_0 \ln 2$$ |
| (Q) | Change in the internal energy of the gas in the process JK | (2) | $$0$$ |
| (R) | Heat given to the gas in the process KL | (3) | $$3RT_0$$ |
| (S) | Change in the internal energy of the gas in the process MJ | (4) | $$-2RT_0 \ln 2$$ |
| (5) | $$-3RT_0 \ln 2$$ |
The diagram (not shown here) is a rectangle in the $$P$$-$$T$$ plane with the numerical labels
$$J:(P_0,T_0),\;K:(P_0,3T_0),\;L:(2P_0,3T_0),\;M:(2P_0,T_0).$$
One mole of a monatomic ideal gas moves along the four sides JK, KL, LM, MJ in that order.
For one mole the ideal-gas equation is $$PV = RT$$ and the internal energy is $$U = \tfrac32 RT.$$ Work done by the gas in any step is $$W = \int P\,dV,$$ and the first law gives $$Q = \Delta U + W.$$ We treat every leg separately.
Case 1: JK (isobaric at $$P_0$$)Temperatures: $$T_0 \rightarrow 3T_0 \;(\Delta T = 2T_0).$$
Change in internal energy
$$\Delta U_{JK} = \tfrac32 R\,(2T_0) = 3RT_0.$$
Volume rises from $$V_J = \frac{RT_0}{P_0}$$ to $$V_K = \frac{3RT_0}{P_0}.$$ Work done $$W_{JK} = P_0\,(V_K-V_J) = P_0 \left(\frac{2RT_0}{P_0}\right)=2RT_0.$$
Case 2: KL (isothermal at $$T = 3T_0$$)Pressure $$P_0 \rightarrow 2P_0,$$ so volume $$V_K = \frac{3RT_0}{P_0}$$ compresses to $$V_L = \frac{3RT_0}{2P_0} = \tfrac12 V_K.$$ Work done $$W_{KL}=RT\ln\!\left(\frac{V_L}{V_K}\right)=3RT_0\ln\!\left(\tfrac12\right)=-3RT_0\ln2.$$
Because the temperature is constant, $$\Delta U_{KL}=0,$$ hence $$Q_{KL}=W_{KL}=-3RT_0\ln2.$$
Case 3: LM (isobaric at $$2P_0$$)Temperature $$3T_0 \rightarrow T_0 \;(\Delta T = -2T_0).$$
Change in internal energy
$$\Delta U_{LM}=\tfrac32 R(-2T_0)=-3RT_0.$$
Volumes: $$V_L=\frac{3RT_0}{2P_0},\;V_M=\frac{RT_0}{2P_0}$$ so $$\Delta V = -\frac{RT_0}{P_0}.$$ Work done $$W_{LM}=2P_0\left(-\frac{RT_0}{P_0}\right)=-2RT_0.$$
Case 4: MJ (isothermal at $$T=T_0$$)Pressure $$2P_0 \rightarrow P_0,$$ volume doubles: $$V_M=\frac{RT_0}{2P_0},\;V_J=\frac{RT_0}{P_0}=2V_M.$$ Work done $$W_{MJ}=RT_0\ln\!\left(\frac{V_J}{V_M}\right)=RT_0\ln2.$$
Again $$\Delta U_{MJ}=0$$ (temperature constant), so $$Q_{MJ}=W_{MJ}=RT_0\ln2.$$
Total work for the cycle$$W_{\text{cycle}} = 2RT_0 + (-3RT_0\ln2) + (-2RT_0) + RT_0\ln2$$ $$\phantom{W_{\text{cycle}}}=0 - 2RT_0\ln2 = -2RT_0\ln2.$$
Now we can match each quantity with List-II:
(P) Work in the entire cycle: $$-2RT_0\ln2$$ → (4)
(Q) $$\Delta U_{JK}=3RT_0$$ → (3)
(R) Heat in KL: $$Q_{KL}=-3RT_0\ln2$$ → (5)
(S) $$\Delta U_{MJ}=0$$ → (2)
Therefore the correct set of matches is P → 4; Q → 3; R → 5; S → 2, which is Option B.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.