Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The energy levels of an atom is shown in figure.
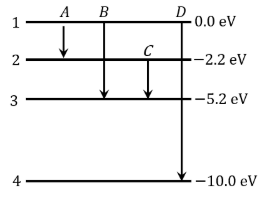
Which one of these transitions will result in the emission of a photon of wavelength 124.1 nm?
Given ($$h = 6.62 \times 10^{-34}$$ J s)
Solution :
Energy of photon emitted is equal to the difference between the energy levels.
Using,
E = hc/λ
Given :
h = 6.62 × 10⁻³⁴ J s
c = 3 × 10⁸ m/s
λ = 124.1 nm = 124.1 × 10⁻⁹ m
Substituting,
E = (6.62 × 10⁻³⁴ × 3 × 10⁸)/(124.1 × 10⁻⁹)
E ≈ 1.6 × 10⁻¹⁸ J
Now,
1 eV = 1.6 × 10⁻¹⁹ J
Therefore,
E = (1.6 × 10⁻¹⁸)/(1.6 × 10⁻¹⁹)
= 10 eV
From the figure, transition from 0 eV to −10 eV has energy difference of 10 eV.
Hence, transition D will emit photon of wavelength 124.1 nm.
Final Answer :
D
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
