Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The variation of stopping potential $$V_0$$ as a function of the frequency $$(\nu)$$ of the incident light for a metal is shown in figure. The work function of the surface is
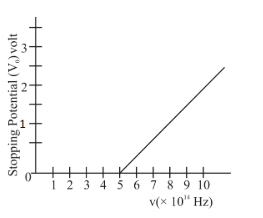
We need to find the work function of a metal surface from the variation of stopping potential $$V_0$$ as a function of the frequency $$\nu$$ of incident light.
Einstein’s photoelectric equation relates the maximum kinetic energy of emitted electrons to the frequency of incident light:
$$eV_0 = h\nu - \phi$$
Here, $$e$$ is the electron charge, $$V_0$$ is the stopping potential, $$h$$ is Planck’s constant, $$\nu$$ is the frequency of incident light, and $$\phi$$ is the work function of the metal.
Rearranging this equation gives $$V_0 = \frac{h}{e}\nu - \frac{\phi}{e}$$, which is a linear relation in $$\nu$$ with slope $$\frac{h}{e}$$ and y-intercept $$-\frac{\phi}{e}$$.
By examining the graph of $$V_0$$ versus $$\nu$$, the threshold frequency $$\nu_0$$, at which $$V_0 = 0$$, corresponds to the x-intercept. At this point, the work function satisfies:
$$\phi = h\nu_0$$
Using Planck’s constant $$h = 6.63 \times 10^{-34}\,\text{J}\,\text{s}$$ and converting joules to electronvolts via $$1\,\text{eV} = 1.6 \times 10^{-19}\,\text{J}$$, and noting from the graph that $$\nu_0 = 5 \times 10^{14}\,\text{Hz}$$, we calculate:
$$\phi = h\nu_0 = 6.63 \times 10^{-34} \times 5 \times 10^{14} = 33.15 \times 10^{-20}\,\text{J}$$
Converting this energy into electronvolts gives:
$$\phi = \frac{33.15 \times 10^{-20}}{1.6 \times 10^{-19}} = \frac{33.15}{16} \approx 2.07\,\text{eV}$$
Therefore, the work function of the metal surface is 2.07 $$\text{ eV}$$.
Create a FREE account and get:
Educational materials for JEE preparation
