Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
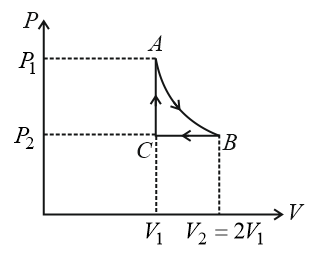
$$n$$ mole of a perfect gas undergoes a cyclic process ABCA (see figure) consisting of the following processes.
$$A \to B$$: Isothermal expansion at temperature $$T$$ so that the volume is doubled from $$V_1$$ to $$V_2 = 2V_1$$ and pressure changes from $$P_1$$ to $$P_2$$
$$B \to C$$: Isobaric compression at pressure $$P_2$$ to initial volume $$V_1$$.
$$C \to A$$: Isochoric change leading to change of pressure from $$P_2$$ to $$P_1$$
Total work done in the complete cycle ABCA is:
We have $$n$$ moles of a perfect gas undergoing a cyclic process ABCA.
For $$A \to B$$ (isothermal expansion at temperature $$T$$ from $$V_1$$ to $$V_2 = 2V_1$$), the work done is $$W_{AB} = nRT\ln\frac{V_2}{V_1} = nRT\ln 2$$.
Since $$A \to B$$ is isothermal, we have $$P_1V_1 = P_2V_2 = nRT$$. With $$V_2 = 2V_1$$, this gives $$P_2 = \frac{P_1}{2}$$ and $$P_2V_1 = \frac{nRT}{2}$$.
For $$B \to C$$ (isobaric compression at pressure $$P_2$$ from $$V_2 = 2V_1$$ back to $$V_1$$), the work done is $$W_{BC} = P_2(V_1 - V_2) = P_2(V_1 - 2V_1) = -P_2V_1 = -\frac{nRT}{2}$$.
For $$C \to A$$ (isochoric process at constant volume $$V_1$$), no work is done since volume does not change. So $$W_{CA} = 0$$.
The total work done in the complete cycle is $$W = W_{AB} + W_{BC} + W_{CA} = nRT\ln 2 - \frac{nRT}{2} + 0 = nRT\left(\ln 2 - \frac{1}{2}\right)$$.
Hence, the correct answer is Option A.
Create a FREE account and get:
Educational materials for JEE preparation
