NTA JEE Main 24th February 2021 Shift 1
For the following questions answer them individually
NTA JEE Main 24th February 2021 Shift 1 - Question 1
The work done by a gas molecule in an isolated system is given by, $$W = \alpha \beta^2 e^{-\frac{x^2}{\alpha k T}}$$, where $$x$$ is the displacement, $$k$$ is the Boltzmann constant and $$T$$ is the temperature. $$\alpha$$ and $$\beta$$ are constants. Then the dimensions of $$\beta$$ will be:
NTA JEE Main 24th February 2021 Shift 1 - Question 2
If the velocity-time graph has the shape AMB, what would be the shape of the corresponding acceleration-time graph?
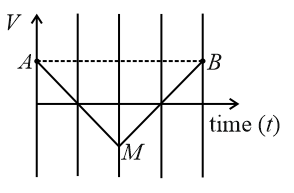
NTA JEE Main 24th February 2021 Shift 1 - Question 3
Moment of inertia M.I. of four bodies, having same mass and radius, are reported as;
$$I_1$$ = M.I. of thin circular ring about its diameter,
$$I_2$$ = M.I. of circular disc about an axis perpendicular to disc and going through the centre,
$$I_3$$ = M.I. of solid cylinder about its axis and
$$I_4$$ = M.I. of solid sphere about its diameter.
Then:
NTA JEE Main 24th February 2021 Shift 1 - Question 4
Consider two satellites $$S_1$$ and $$S_2$$ with periods of revolution 1hr and 8hr respectively revolving around a planet in circular orbits. The ratio of angular velocity of satellite $$S_1$$ to the angular velocity of satellite $$S_2$$ is:
NTA JEE Main 24th February 2021 Shift 1 - Question 5
Four identical particles of equal mass of 1 kg are made to move along the circumference of a circle of radius 1 m under the action of their own mutual gravitational attraction. The speed of each particle will be:
NTA JEE Main 24th February 2021 Shift 1 - Question 6
Two stars of masses $$m$$ and $$2m$$ at a distance $$d$$ rotate about their common centre of mass in free space. The period of revolution is:
NTA JEE Main 24th February 2021 Shift 1 - Question 7
If $$Y$$, $$K$$ and $$\eta$$ are the values of Young's modulus, bulk modulus and modulus of rigidity of any material respectively. Choose the correct relation for these parameters.
NTA JEE Main 24th February 2021 Shift 1 - Question 8
Each side of a box made of metal sheet in cubic shape is $$a$$ at room temperature $$T$$, the coefficient of linear expansion of the metal sheet is $$\alpha$$. The metal sheet is heated uniformly, by a small temperature $$\Delta T$$, so that its new temperature is $$T + \Delta T$$. Calculate the increase in the volume of the metal box.
NTA JEE Main 24th February 2021 Shift 1 - Question 9
Match List I with List II.
| List I | List II |
|---|---|
| (a) Isothermal | (i) Pressure constant |
| (b) Isochoric | (ii) Temperature constant |
| (c) Adiabatic | (iii) Volume constant |
| (d) Isobaric | (iv) Heat content is constant |
Choose the correct answer from the options given below:
NTA JEE Main 24th February 2021 Shift 1 - Question 10
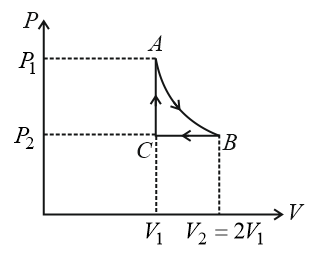
$$n$$ mole of a perfect gas undergoes a cyclic process ABCA (see figure) consisting of the following processes.
$$A \to B$$: Isothermal expansion at temperature $$T$$ so that the volume is doubled from $$V_1$$ to $$V_2 = 2V_1$$ and pressure changes from $$P_1$$ to $$P_2$$
$$B \to C$$: Isobaric compression at pressure $$P_2$$ to initial volume $$V_1$$.
$$C \to A$$: Isochoric change leading to change of pressure from $$P_2$$ to $$P_1$$
Total work done in the complete cycle ABCA is:
.webp)


