Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Following figure shows two processes A and B for a gas. If $$\Delta Q_A$$ and $$\Delta Q_B$$ are the amount of heat absorbed by the system in two cases, and $$\Delta U_A$$ and $$\Delta U_B$$ are changes in internal energies, respectively, then:
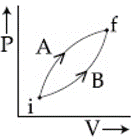
Based on the First Law of Thermodynamics and the properties of state functions, the solution is as follows:
1. Internal Energy Change ($$\Delta U$$):
Internal energy is a state function, meaning it only depends on the initial and final states of the system, not the path taken. Since both processes A and B start and end at the same points on the P-V diagram:
$$\Delta U_A = \Delta U_B$$
2. Work Done (W):
Work done in a thermodynamic process is equal to the area under the curve on a P-V diagram.
3. Heat Absorbed ($$\Delta Q$$):
According to the First Law of Thermodynamics:
$$\Delta Q = \Delta U + W$$
Applying this to both processes:
Since $$\Delta U$$ is the same for both but $$W_A > W_B$$, it follows that:
$$\boxed{\Delta Q_A > \Delta Q_B \quad \text{and} \quad \Delta U_A = \Delta U_B}$$
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.