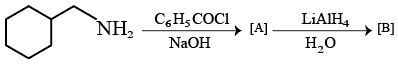NTA JEE Mains 22nd Jan 2026 Shift 2
For the following questions answer them individually
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 51
Identify the correct statements
A. Hydrated salts can be used as primary standard.
B. Primary standard should not undergo any reaction with air.
C. Reactions of primary standard with an other substance should be instantaneous and stoichiometric.
D. Primary standard should not be soluble in water.
E. Primary standard should have low relative molar mass.
Choose the correct answer from th e options given below :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 52
When 1 g of cmnpow1d (X) is subjected to Kjeldahl's method for estimation of nitrogen, 15 mL 1 M $$H_{2}SO_{4}$$ was neutrilized by ammonia evolved. The percentage of nitrogen in compound
(X) is :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 53
Given below are two statements :
Statement I : C < 0 < N < F is the correct order in terms of first ionization enthalpy values.
Statement II: S > Se > Te > Po > 0 is the correct order in terms of the magnitude of electron gain enthalpy values.
ln the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 54
The IUPAC name of the following compound is :
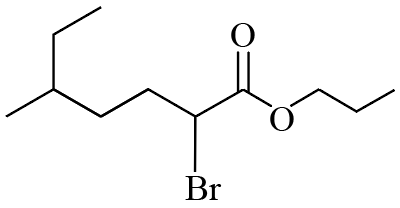
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 55
$$A + 2B \rightarrow AB_{2}$$
36.0 g of ' A' (Molar mass : 60 g $$mol^{-1}$$) and 56.0 g of 'B' (Molar mass: 80 g $$mol^{-1}$$) are allowed to react. Which of the following statements are correct?
A. 'A' is the limiting reagent.
B. 77.0 g of $$AB_{2}$$ is formed.
C. Molar mass of $$AB_{2} is 140 g $$mol^{-1}$$
D. 15.0 g of A is left unreacted after the completion of reaction.
Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 57
The dibromo compound [P] (molecular formula : $$C_{9}H_{10}Br_{2}$$) when heated with excess sodamide followed by treatment with dilute HO gives [Q]. On wanning [Q] with mercuric sulphate and
dilute sulphuric acid yield [R] which gives positive Iodofonn test but negative Tollen's test. The compound [P] is:
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 58
Match List - I with List - II.

choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 59
Given below are two statements:
Statement I : Elements 'X' and 'Y' are the most and least electronegative elements, respectively among N, As, Sb and P. The nature of the oxides $$X_{2}O_{3}$$ and $$y_{2}O_{3}$$ is acidic and
amphoteric, respectively.
Statement II:$$BCl_{3}$$ is covalent in nature and gets hydrolysed in water. It produces $$[B(OH)_{4}]^{-}$$ and $$[B(H_{2}O)_{6}]^{3+}$$ in aqueous medium.
In the Light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 60
At T(K), 100 g of 98% $$H_{2}SO_{4}$$ (w /w) aqueous solution is mixed with 100 g of 49% $$H_{2}SO_{4}$$ (w /w) aqueous solution. What is the mole fraction of $$H_{2}SO_{4}$$ in the resultant solution?
(Given: Atomic mass H=1 u ; s=32 u ; 0 = 16 u).
(Assume that temperature after mixing remains constant)