NTA JEE Mains 22nd Jan 2026 Shift 2
For the following questions answer them individually
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 61
Which of the followmg mixture gives a buffer solution with pH=9.25?
Given : $$pK_{b}$$ $$(NH_{4}OH)$$ = 4.75
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 62
Consider the following reduction processes :
$$Al^{3+} + 3e^{-} \rightarrow Al(s), E^{\circ} = -1.66V$$
$$Fe^{3+} + e^{-} \rightarrow Fe^{2+}, E^{\circ} = +0.77V$$
$$Co^{3+} + e^{-} \rightarrow Co^{2+}, E^{\circ} = +1.81V$$
$$Cr^{3+} + 3e^{-} \rightarrow Cr(s), E^{\circ} = -0.74V$$
The tendency to act as reducing agent decreases in the order :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 63
The compound A, $$C_{8}H_{8}O_{2}$$ reacts with acetophenone to form a single product via cross-Aldol condensation. The compound A on reaction with cone. NaOH forms a substituted benzyl alcohol as one of the two products. The compound A is :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 64
Given below are two statements :
Statement I : The first ionization enthalpy of Cr is lower than that of Mn.
Statement II : The second and third ionization enthalpies of Cr are higher than those of Mn. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 65
Among $$H_{2}S$$, $$H_{2}O$$, $$NF_{3}$$, $$NH_{3}$$ and $$CHC1_{3}$$, identify the molecule (X) with lowest dipole moment value. The number of lone pairs of electrons present on the central atom of the molecule (X) is :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 66
3, 3-Dimethyl-2-butanol cannot be prepared by:
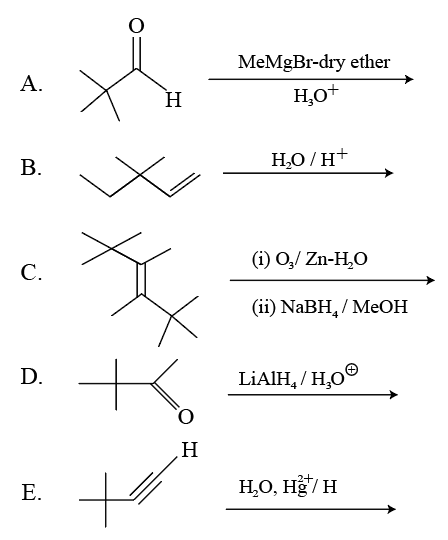
Choose the correct answer from the options given below:
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 67
$$[Ni (PPh_{3})_{2}Cl_{2} ]$$ is a paramagnetic complex. Identify the INCORRECT statements about this complex.
A. The complex exhibits geometrical isomerism.
B. The complex is white in colour.
C. The calculated spin-only magnetic moment of the complex is 2.84 BM.
D. The calculated CFSE (Crystal Field Stabilization Energy) of Ni in this complex is $$-0.8 \triangle_{0}$$
E. The geometrical arrangement of ligands in this complex is similar to that in $$Ni(CO)_{4}$$.
Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 68
Correct statements regarding Arrhenius equation among the following are :
A. Factor $$e^{-Ea / RT}$$ corresponds to fraction of molecules having kinetic energy less than Ea.
B. At a given temperature, lower the Ea, faster is the reaction.
C. Increase in temperature by about $$10^{\circ}C$$ doubles the rate of reaction.
D. Plot of log k vs $$\frac{1}{T}$$ gives a straight line with slope = $$- \frac{Ea}{R}$$.
Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 69
The energy of first (lowest) Balmer line of H atom is x J. The energy (in J) of second Balmer line of H atom is :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Question 70
Consider the following reaction :
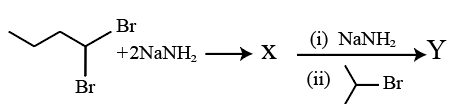
The product Y formed is :


