Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
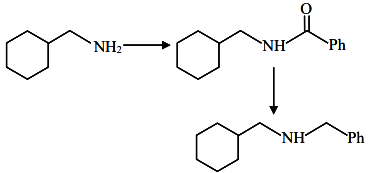
Given compound: Cyclohexyl-CH₂-NH₂ (a primary amine)
This is Schotten-Baumann Reaction (Acylation of amines)
Cyclohexyl-CH₂-NHCOPh
(N-benzoyl derivative / amide)
This is Reduction of amides using LiAlH₄
So:
-NHCOC₆H₅ → -NHCH₂C₆H₅
Cyclohexyl-CH₂-CH₂-NH-CH₂-Ph
(This is a secondary amine)
B = Cyclohexyl-CH₂-NH-CH₂-Ph
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
