NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 51
Identify the correct statements
A. Hydrated salts can be used as primary standard.
B. Primary standard should not undergo any reaction with air.
C. Reactions of primary standard with an other substance should be instantaneous and stoichiometric.
D. Primary standard should not be soluble in water.
E. Primary standard should have low relative molar mass.
Choose the correct answer from th e options given below :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 52
When 1 g of cmnpow1d (X) is subjected to Kjeldahl's method for estimation of nitrogen, 15 mL 1 M $$H_{2}SO_{4}$$ was neutrilized by ammonia evolved. The percentage of nitrogen in compound
(X) is :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 53
Given below are two statements :
Statement I : C < 0 < N < F is the correct order in terms of first ionization enthalpy values.
Statement II: S > Se > Te > Po > 0 is the correct order in terms of the magnitude of electron gain enthalpy values.
ln the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 54
The IUPAC name of the following compound is :
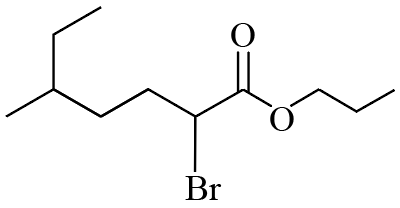
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 55
$$A + 2B \rightarrow AB_{2}$$
36.0 g of ' A' (Molar mass : 60 g $$mol^{-1}$$) and 56.0 g of 'B' (Molar mass: 80 g $$mol^{-1}$$) are allowed to react. Which of the following statements are correct?
A. 'A' is the limiting reagent.
B. 77.0 g of $$AB_{2}$$ is formed.
C. Molar mass of $$AB_{2} is 140 g $$mol^{-1}$$
D. 15.0 g of A is left unreacted after the completion of reaction.
Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 56
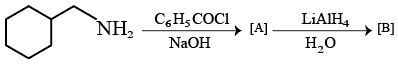
The final product [B] is :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 57
The dibromo compound [P] (molecular formula : $$C_{9}H_{10}Br_{2}$$) when heated with excess sodamide followed by treatment with dilute HO gives [Q]. On wanning [Q] with mercuric sulphate and
dilute sulphuric acid yield [R] which gives positive Iodofonn test but negative Tollen's test. The compound [P] is:
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 58
Match List - I with List - II.

choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 59
Given below are two statements:
Statement I : Elements 'X' and 'Y' are the most and least electronegative elements, respectively among N, As, Sb and P. The nature of the oxides $$X_{2}O_{3}$$ and $$y_{2}O_{3}$$ is acidic and
amphoteric, respectively.
Statement II:$$BCl_{3}$$ is covalent in nature and gets hydrolysed in water. It produces $$[B(OH)_{4}]^{-}$$ and $$[B(H_{2}O)_{6}]^{3+}$$ in aqueous medium.
In the Light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 60
At T(K), 100 g of 98% $$H_{2}SO_{4}$$ (w /w) aqueous solution is mixed with 100 g of 49% $$H_{2}SO_{4}$$ (w /w) aqueous solution. What is the mole fraction of $$H_{2}SO_{4}$$ in the resultant solution?
(Given: Atomic mass H=1 u ; s=32 u ; 0 = 16 u).
(Assume that temperature after mixing remains constant)
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 61
Which of the followmg mixture gives a buffer solution with pH=9.25?
Given : $$pK_{b}$$ $$(NH_{4}OH)$$ = 4.75
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 62
Consider the following reduction processes :
$$Al^{3+} + 3e^{-} \rightarrow Al(s), E^{\circ} = -1.66V$$
$$Fe^{3+} + e^{-} \rightarrow Fe^{2+}, E^{\circ} = +0.77V$$
$$Co^{3+} + e^{-} \rightarrow Co^{2+}, E^{\circ} = +1.81V$$
$$Cr^{3+} + 3e^{-} \rightarrow Cr(s), E^{\circ} = -0.74V$$
The tendency to act as reducing agent decreases in the order :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 63
The compound A, $$C_{8}H_{8}O_{2}$$ reacts with acetophenone to form a single product via cross-Aldol condensation. The compound A on reaction with cone. NaOH forms a substituted benzyl alcohol as one of the two products. The compound A is :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 64
Given below are two statements :
Statement I : The first ionization enthalpy of Cr is lower than that of Mn.
Statement II : The second and third ionization enthalpies of Cr are higher than those of Mn. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 65
Among $$H_{2}S$$, $$H_{2}O$$, $$NF_{3}$$, $$NH_{3}$$ and $$CHC1_{3}$$, identify the molecule (X) with lowest dipole moment value. The number of lone pairs of electrons present on the central atom of the molecule (X) is :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 66
3, 3-Dimethyl-2-butanol cannot be prepared by:
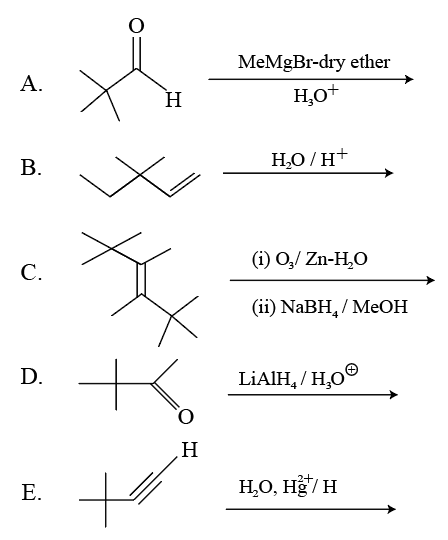
Choose the correct answer from the options given below:
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 67
$$[Ni (PPh_{3})_{2}Cl_{2} ]$$ is a paramagnetic complex. Identify the INCORRECT statements about this complex.
A. The complex exhibits geometrical isomerism.
B. The complex is white in colour.
C. The calculated spin-only magnetic moment of the complex is 2.84 BM.
D. The calculated CFSE (Crystal Field Stabilization Energy) of Ni in this complex is $$-0.8 \triangle_{0}$$
E. The geometrical arrangement of ligands in this complex is similar to that in $$Ni(CO)_{4}$$.
Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 68
Correct statements regarding Arrhenius equation among the following are :
A. Factor $$e^{-Ea / RT}$$ corresponds to fraction of molecules having kinetic energy less than Ea.
B. At a given temperature, lower the Ea, faster is the reaction.
C. Increase in temperature by about $$10^{\circ}C$$ doubles the rate of reaction.
D. Plot of log k vs $$\frac{1}{T}$$ gives a straight line with slope = $$- \frac{Ea}{R}$$.
Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 69
The energy of first (lowest) Balmer line of H atom is x J. The energy (in J) of second Balmer line of H atom is :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 70
Consider the following reaction :
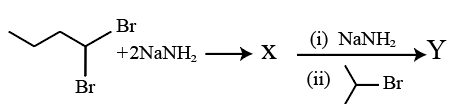
The product Y formed is :
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 71
Consider the following electrochemical cell : $$Pt| O_{2}(g)(1 bar) | HCl(aq)||M^{2+}(aq$$, $$1.0M)|M(s)$$
The pH above which , oxygen gas would start to evolve at anode is ____ (nearest integer).
[Given: $$E_{M^{2+}/m}^\circ = 0.994V$$ , $$E_{O^{2}/H_{2}O}^\circ = 1.23V$$ are standard reduction potential and $$\frac{RT}{F}(2.303)=0.059V$$ at the given condition]
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 72
Consider $$A \xrightarrow{k_1} B$$ and $$ C \xrightarrow{k_2} D$$ are two reactions. If the rate constant ($$k_{1}$$) of the $$A \rightarrow B$$ reaction can be expressed by the followmg equation $$\log_{10}K = 14.34- \frac{1.5 \times 10^{4}}{T/K}$$ and activation energy $$C\rightarrow D$$ reaction ($$Ea_{2}$$) is $$\frac{1}{5}th$$ of the $$A\rightarrow B$$ reaction ($$Ea_{1}$$), then the value of ($$Ea_{2}$$) is _____________kJ $$mol^{-1}$$. (Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 73
Among the following oxides of 3d elements, the number of mixed oxides are ___________.
$$Ti_{2}O_{3}$$, $$V_{2}O_{4}$$, $$Cr_{2}O_{3}$$, $$Mn_{3}O_{4}$$, $$Fe_{3}O_{4}$$, $$Fe_{2}O_{3}$$, $$Co_{3}O_{4}$$
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 74
If the enthalpy of sublimation of Li is 155 kj $$mol^{-1}$$, enthalpy of dissociation of $$F_{2}$$ is 150 kj $$mol^{-1}$$, ionization enthalpy of Li is 520 kj $$mol^{-1}$$, electron gain enthalpy of F is - 313 kj $$mol^{-1}$$, standard enthalpy of formation of LiF is - 594 kj $$mol^{-1}$$. The magnitude of lattice enthalpy of LiF is _________ kJ $$mol_{-1}$$. (Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2026 Shift 2 - Chemistry - Question 75
The mass of benzanilide obtained from the benzoylation reaction of 5.8 g of aniline, if yield of product is 82%, is ____ g (nearest integer) .
(Given molar mass in g $$mol^{-1}$$ H : 1, C: 12, N: 14, O : 16)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




