NTA JEE Mains 22nd Jan 2026 Shift 1
For the following questions answer them individually
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 61
A 'p'-block element (E) and hydrogen form a binary cation $$(EH_{x})^{+}$$ , while $$EH_{3}$$ on treatment with $$K_{2}HgI_{4}$$ in alkaline medium gives a precipitate of basic mercury(II)amido- iodine. Given below are first ionisation enthalpy values ($$kJ mol^{-1}$$) for first element each from group 13, 14, 15 and 16. Identify the correct first ionisation enthalpy value for element E.
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 62
Given below are two statements:
Statement I: Benzene is nitrated to give nitrobenzene, which on further treatment
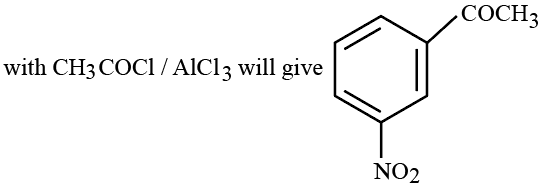
Statement II: -$$NO_{2}$$ group is a m-directing, and deactivating group.
In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 63
Given below are two statements:
Statement I: Phenol on treatment with . $$CHCL_{3}$$/aq. $$KOH$$ under refluxing condition, followed by acidification produces $$p$$-hydroxy benzaldehyde as the major product and $$o$$-hydroxy benzaldehyde as the minor product.
Statement II: The mixture of $$p$$-hydroxybenzaldehyde and $$o$$-
hydroxybenzaldehyde can be easily separated through steam distillation.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 64
A first row transition metal (M) does not liberate $$H_{2}$$ gas from dilute HCI. 1 mol of aqueous solution of $$MSO_{4}$$ is treated with excess of aqueous KCN and then $$H_{2} S(g)$$ is passed through the solution. The amount of MS (metal sulphide) formed from the above reaction is _______ mol.
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 65
Consider the transition metal ions $$Mn^{3+}, Cr^{3+}, Fe ^{3+}$$ and $$Co^{3+}$$ and all form low spin octahedral complexes. The correct decreasing order of unpaired electrons in their respective d-orbitals of the complexes is
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 66
Two p-block elements $$X$$ and $$Y$$ form fluorides of the type $$EF_{3}$$. The fluoride compound $$XF_{3}$$ is a Lewis acid and $$YF_{3}$$ is a Lewis base. The hybridizations of the central atoms of $$XF_{3}$$ and $$YF_{3}$$ respectively are
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 67
'A' is a neutral organic compound $$(M.F : C_{8}H_{9}ON)$$. On treatment with aqueous $$Br_{2}/Ho_{(-)}$$, 'A' forms a compound 'B' which is soluble in dilute acid. 'B' on treatment with aqueous $$NaNO_{2} / HCl (0-5^{o}C)$$ produces a compound 'C' which on treatment with $$CuCN/NaCN$$ produces 'D'. Hydrolysis of 'D' produces 'E' which is also obtainable from the hydrolysis of'A'. 'E' on treatment with acidified $$KMnO_{4}$$ produces 'F'. 'F' contains two different types of hydrogen atoms. The structure of 'A' is
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 68
Match the LIST-I with LIST-II
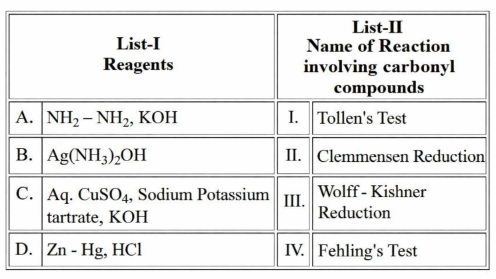
Choose the correct answer from the options given below:
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 69
In the reaction,
$$ 2Al(s)+6HCl(aq)\rightarrow2Al^{3+}(aq)+6cl^{-}(aq)+3H_{2}(g)$$
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 70
The correct order of reactivity of $$CH_{3} Br$$ in methanol with the following
nucleophiles is
$$F^{-}, I^{-}, C_{2}H_{5}O^{-}$$ and $$C_{6}H_{5}O^{-}$$


