NTA JEE Mains 22nd Jan 2026 Shift 1
For the following questions answer them individually
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 51
Given below are two statements:
Statement I: Sucrose is dextrorotatory. However, sucrose upon hydrolysis gives a solution having mixture of products. This solution shows laevorotation.
Statement II : Hydrolysis of sucrose gives glucose and fructose. Since the laevorotation of glucose is more than the dextrorotation of fructose, the resulting solution becomes laevorotato1y.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 52
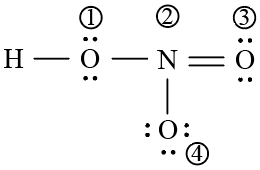
The formal charges on the atoms marked as (1) to (4) in the Lew is representation
of $$HNO_{3}$$ molecule respectively are
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 53
Consider a solution of $$CO_{2} (g)$$ dissolved in water in a closed container.
Which one of the following plots correctly represents variation of log (partial pressure of $$CO_{2}$$ in vapour phase above water) [y-axis] with log (mole fraction of $$CO_{2}$$ in water) [x-axis] at $$ 25^{o}C $$?
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 54
The energy required by electrons, present in the first Bohr orbit of hydrogen atom to J $$ mol^{-1}C $$ be excited to second Bohr orbit is ______ .
Given $$ R_{H}=2.18\times 10^{-11} $$
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 55
Match the LIST-I with LIST-II
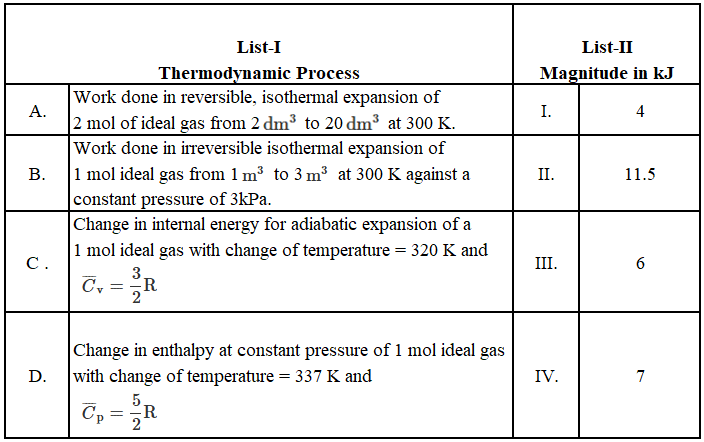
Choose the correct answer from the options given below:
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 56
The correct order of the rate of reaction of the following reactants with nucleophile by $$S_{N}1$$ mechanism is :
(Given: Structures I and II are rigid)
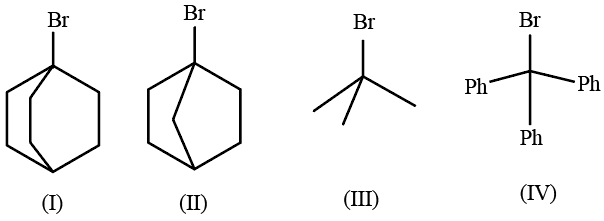
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 57
Given below are two statements:
Statement I: The Henry's law constant $$K_{H}$$ is constant with respect to variations in solution's concentration over the range for which the solution is ideally dilute.
Statement II: $$K_{H}$$ does not differ for the same solute in different solvents.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 58
As compared with chlorocyclohexane, which of the following statements correctly apply to chlorobenzene?
A. The magnitude of negative charge is more on chlorine atom.
B. The C - Cl bond has partial double bond character.
C. C - Cl bond is less polar.
D. C - Cl bond is longer due to repulsion between delocalised electrons of the aromatic ring and lone pairs of electrons of chlorine.
E. The C - Cl bond is formed using $$ sp^{2} $$ hybridised orbital of carbon.
Choose the correct answer from the options given below:
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 59
A$$\rightarrow$$ product (First order reaction).
Three sets of experiment were performed for a reaction under similar experimental conditions:
Run 1 $$\Rightarrow$$ 100 mL of 10 M solution of reactant A
Run 2 $$\Rightarrow$$ 200 mL of 10 M solution of reactant A
Run 3 $$\Rightarrow$$ 100 mL of 10 M solution of reactant A + 100 mL of $$H_{2}O$$ added.
The correct variation of rate of reaction is
NTA JEE Mains 22nd Jan 2026 Shift 1 - Question 60
Given below are two statements:
Statement I: The halogen that makes longest bond with hydrogen in HX, has the smallest covalent radius in its group.
Statement II: A group 15 element's hydride $$EH_{3}$$ has the lowest boiling point among corresponding hydrides of other group 15 elements. The maximum covalency of that element E is 4.
In the light of the above statements, choose the correct answer from the options given below


