Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
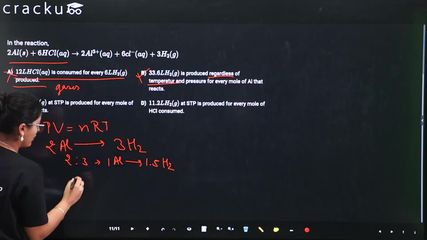
In the reaction,
$$ 2Al(s)+6HCl(aq)\rightarrow2Al^{3+}(aq)+6cl^{-}(aq)+3H_{2}(g)$$
In the reaction,
2Al (s) + 6HCl (aq) → 2$$Al^{+3}$$ (aq) + 6$$Cl^-$$ (aq) + 3$$H_2$$ (g)
(a) 6L HCl(aq) is consumed for every 3L $$H_2$$(g) produced
(b) This is incorrect statement because according to ideal gas equation (PV = nRT) volume of $$H_2$$ produces will depend on temperature and pressure.
(c) 67 .2 L $$H_2$$ (g) at STP (standard Temperature and Pressure, T = 373.15K, P = 1atm) is produced for every mole Al that reacts
(d)11.2 L $$H_2$$(g) at STP is produced for every mole HCl(aq) consumed
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
