NTA JEE Main 9th January 2020 Shift 1
For the following questions answer them individually
NTA JEE Main 9th January 2020 Shift 1 - Question 21
The distance $$x$$ covered by a particle in one dimensional motion varies with time $$t$$ as $$x^2 = at^2 + 2bt + c$$. If the acceleration of the particle depends on $$x$$ as $$x^{-n}$$, where $$n$$ is an integer, the value of $$n$$ is ___________.
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 1 - Question 22
One end of a straight uniform $$1 \; m$$ long bar is pivoted on horizontal table. It is released from rest when it makes an angle $$30°$$ from the horizontal (see figure). Its angular speed when it hits the table is given as $$\sqrt{n}$$ rad $$s^{-1}$$, where $$n$$ is an integer. The value of $$n$$ is ___________.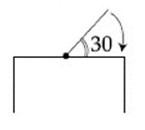
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 1 - Question 23
A body of mass $$m = 10$$ kg is attached to one end of a wire of length $$0.3$$ m. What is the maximum angular speed (in rad $$s^{-1}$$) with which it can be rotated about its other end in a space station without breaking the wire?
[Breaking stress of wire $$(\sigma) = 4.8 \times 10^7$$ N $$m^{-2}$$ and area of cross-section of the wire $$= 10^{-2}$$ $$cm^2$$]
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 1 - Question 24
In a fluorescent lamp choke (a small transformer) $$100V$$ of reverse voltage is produced when the choke current changes uniformly from $$0.25A$$ to $$0$$ in a duration of $$0.025 \; ms$$. The self-inductance of the choke (in $$mH$$) is estimated to be ___________.
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 1 - Question 25
Both the diodes used in the circuit shown are assumed to be ideal and have negligible resistance when these are forward biased. Built in potential in each diode is $$0.7V$$. For the input voltages shown in the figure, the voltage (in Volts) at point A is ___________.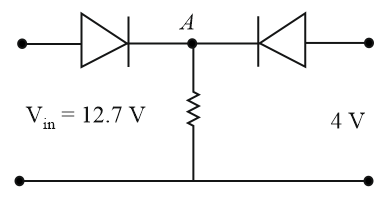
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 1 - Question 26
The de Broglie wavelength of an electron in the $$4^{th}$$ Bohr orbit is:
NTA JEE Main 9th January 2020 Shift 1 - Question 27
B has a smaller first ionization enthalpy than Be. Consider the following statement:
(I) it is easier to remove 2p electron than 2s electron
(II) 2p electron of B is more shielded from the nucleus by the inner core of electrons than the 2s electrons of Be
(III) 2s electron has more penetration power than 2p electron
(IV) atomic radius of B is more than Be
(atomic number B : 5, Be = 4)
The correct statements are:
NTA JEE Main 9th January 2020 Shift 1 - Question 28
The acidic, basic and amphoteric oxides, respectively, are:
NTA JEE Main 9th January 2020 Shift 1 - Question 29
If the magnetic moment of a di-oxygen species is 1.73 B.M., it may be:
NTA JEE Main 9th January 2020 Shift 1 - Question 30
If enthalpy of atomization for $$Br_2(l)$$ is $$x$$ kJ/mol and bond enthalpy for $$Br_2$$ is $$y$$ kJ/mol, the relation between them:


