NTA JEE Main 9th January 2020 Shift 1
For the following questions answer them individually
NTA JEE Main 9th January 2020 Shift 1 - Question 31
The $$K_{sp}$$ for the following dissociation is $$1.6 \times 10^{-5}$$
$$PbCl_{2(s)} \rightleftharpoons Pb^{2+}_{(aq)} + 2Cl^{-}_{(aq)}$$
Which of the following choices is correct for a mixture of 300 mL 0.134 M $$Pb(NO_3)_2$$ and 100 mL 0.4 M NaCl?
NTA JEE Main 9th January 2020 Shift 1 - Question 32
The compound that cannot act both as oxidizing and reducing agent is:
NTA JEE Main 9th January 2020 Shift 1 - Question 33
'X' melts at low temperature and is a bad conductor of electricity in both liquid and solid state. X is:
NTA JEE Main 9th January 2020 Shift 1 - Question 34
The increasing order of basicity for the following intermediates is (from weak to strong):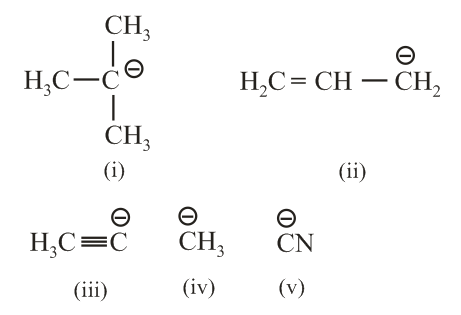
NTA JEE Main 9th January 2020 Shift 1 - Question 35
The correct order of heat of combustion for following alkadienes is: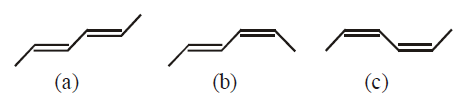
NTA JEE Main 9th January 2020 Shift 1 - Question 36
The major product (Y) in the following reactions is:
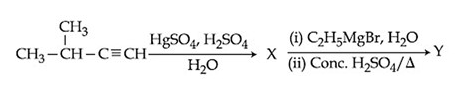
NTA JEE Main 9th January 2020 Shift 1 - Question 37
For the following reactions:
$$A \xrightarrow{700K} Product$$
$$A \xrightarrow[catalyst]{500K} Product$$
It was found that the $$E_a$$ is decreased by 30 KJ/mol in the presence of catalyst. If the rate remains unchanged, the activation energy for catalysed reaction is (Assume pre-exponential factor is same):
NTA JEE Main 9th January 2020 Shift 1 - Question 38
According to the following diagram, A reduces $$BO_2$$ when the temperature is: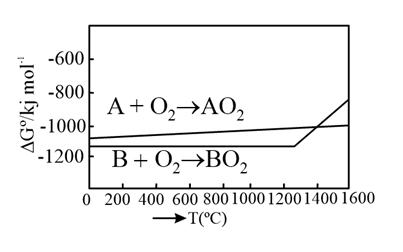
NTA JEE Main 9th January 2020 Shift 1 - Question 39
The electronic configurations of bivalent europium and trivalent cerium are:
(atomic number: Xe = 54, Ce = 58, Eu = 63)
NTA JEE Main 9th January 2020 Shift 1 - Question 40
Complex X of composition $$Cr(H_2O)_6Cl_n$$ has a spin only magnetic moment of 3.83 B.M. It reacts with $$AgNO_3$$ and shows geometrical isomerism. The IUPAC nomenclature of X is:


