NTA JEE Main 9th January 2020 Shift 1
For the following questions answer them individually
NTA JEE Main 9th January 2020 Shift 1 - Question 41
$$[Pd(F)(Cl)(Br)(I)]^{2-}$$ has $$n$$ number of geometrical isomers. Then, the spin-only magnetic moment and crystal field stabilization energy [CFSE] of $$[Fe(CN)_6]^{n-6}$$, respectively, are:
[Note: Ignore the pairing energy]
NTA JEE Main 9th January 2020 Shift 1 - Question 42
Which of these will produce the highest yield Friedel Crafts reaction?
NTA JEE Main 9th January 2020 Shift 1 - Question 43
Identify (A) in the following reaction sequence: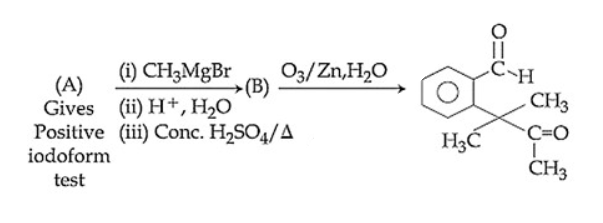
NTA JEE Main 9th January 2020 Shift 1 - Question 44
The major product Z obtained in the following reaction scheme is: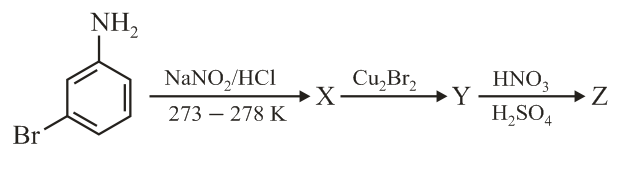
NTA JEE Main 9th January 2020 Shift 1 - Question 45
A chemist has 4 samples of artificial sweetener A, B, C and D. To identify these samples, he performed certain experiments and noted the following observations:
(i) A and D both form blue-violet colour with ninhydrin.
(ii) Lassaigne extract of C gives positive $$AgNO_3$$ test and negative $$Fe_4[Fe(CN)_6]_3$$ test.
(iii) Lassaigne extract of B and D gives positive sodium nitroprusside test.
Based on these observations which option is correct?
NTA JEE Main 9th January 2020 Shift 1 - Question 46
The molarity of $$HNO_3$$ in a sample which has density 1.4 g/mL and mass percentage of 63% is ___________.
(Molecular Weight of $$HNO_3 = 63$$)
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 1 - Question 47
The hardness of a water sample containing $$10^{-3}$$ M $$MgSO_4$$ expressed as $$CaCO_3$$ equivalents (in ppm) is ___________.
(molar mass of $$MgSO_4$$ is 120.37 g/mol)
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 1 - Question 48
How much amount of NaCl should be added to 600 g of water $$(\rho = 1.00 \; g/mL)$$ to decrease the freezing point of water to $$-0.2$$ °C? ___________. (The freezing point depression constant for water $$= 2$$ K $$kg \; mol^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 1 - Question 49
108 g of silver (molar mass 108 $$gmol^{-1}$$) is deposited at cathode from $$AgNO_3(aq)$$ solution by a certain quantity of electricity. The volume (in L) of oxygen gas produced at 273 K and 1 bar pressure from water by the same quantity of electricity is ___________.
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 1 - Question 50
The mass percentage of nitrogen in histamine is ___________.
789
456
123
0.-
Clear All


