NTA JEE Main 27th July 2022 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 31
The correct decreasing order of energy, for the orbitals having following set of quantum numbers:
(A) $$n = 3, l = 0, m = 0$$
(B) $$n = 4, l = 0, m = 0$$
(C) $$n = 3, l = 1, m = 0$$
(D) $$n = 3, l = 2, m = 1$$
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 32
Outermost electronic configurations of four elements A, B, C, D are given below:
(A) $$3s^2$$
(B) $$3s^2 3p^1$$
(C) $$3s^2 3p^3$$
(D) $$3s^2 3p^4$$
The correct order of first ionization enthalpy for them is
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 33
Match List-I with List-II
| List-I | List-II |
|---|---|
| A. $$\Psi_{MO} = \Psi_A - \Psi_B$$ | I. Dipole moment |
| B. $$\mu = Q \times r$$ | II. Bonding molecular orbital |
| C. $$\frac{N_b - N_a}{2}$$ | III. Anti-bonding molecular orbital |
| D. $$\Psi_{MO} = \Psi_A + \Psi_B$$ | IV. Bond order |
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 34
The plot of pH-metric titration of weak base $$NH_4OH$$ vs strong acid $$HCl$$ looks like
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 35
An element A of group 1 shows similarity to an element B belonging to group 2. If A has maximum hydration enthalpy in group 1, then B is
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 36
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Boron is unable to form $$BF_6^{3-}$$
Reason R: Size of B is very small.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 37
Match List-I with List-II
| List-I (Mixture) | List-II (Purification Process) |
|---|---|
| A. Chloroform & Aniline | I. Steam distillation |
| B. Benzoic acid & Naphthalene | II. Sublimation |
| C. Water & Aniline | III. Distillation |
| D. Naphthalene & Sodium chloride | IV. Crystallisation |
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 38
Given below are two statements:
Statement I: The non bio-degradable fly ash and slag from steel industry can be used by cement industry.
Statement II: The fuel obtained from plastic waste is lead free.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 39
Given below are two statements:
Statement I: For KI, molar conductivity increases steeply with dilution.
Statement II: For carbonic acid, molar conductivity increases slowly with dilution.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 40
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Dissolved substances can be removed from a colloidal solution by diffusion through a parchment paper.
Reason R: Particles in a true solution cannot pass through parchment paper but the colloidal particles can pass through the parchment paper.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 41
In neutral or alkaline solution, $$MnO_4^-$$ oxidises thiosulphate to
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 42
Low oxidation state of metals in their complexes are common when ligands
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 43
$$Fe^{3+}$$ cation gives a prussian blue precipitate on addition of potassium ferrocyanide solution due to the formation of
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 44
The structure of A in the given reaction is
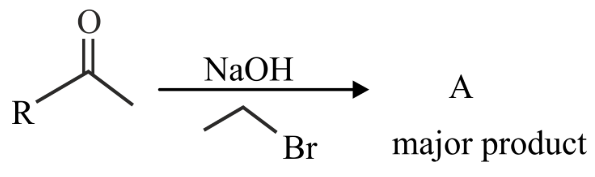
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 45
Major product 'B' of the following reaction sequence is
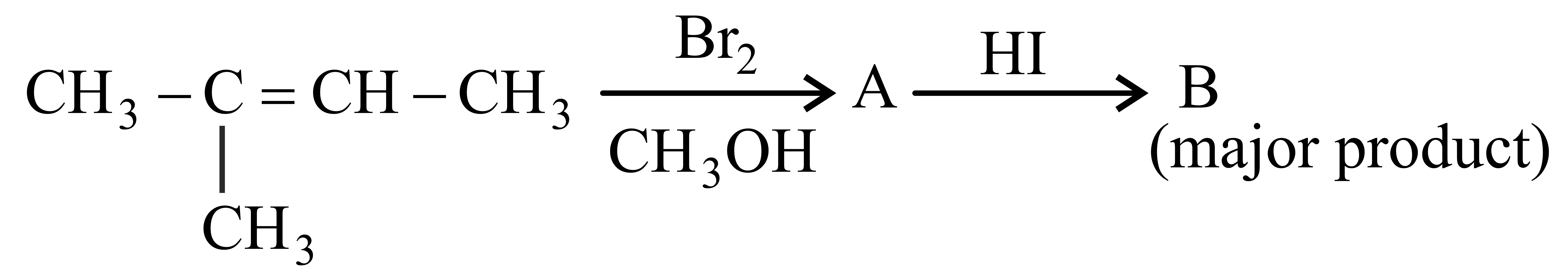
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 46
Match List-I with List-II.
| List-I | List-II |
|---|---|
A
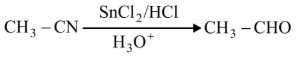 | I. Gatterman Koch reaction |
B
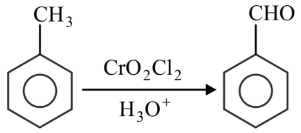 | II. Etard reaction |
C
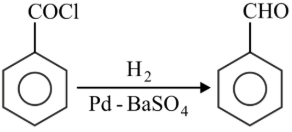 | III. Stephen reaction |
D
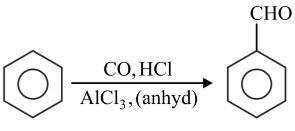 | IV. Rosenmund reaction |
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 47
An organic compound 'A' contains nitrogen and chlorine. It dissolves readily in water to give a solution that turns litmus red. Titration of compound 'A' with standard base indicates that the molecular weight of 'A' is $$131 \pm 2$$. When a sample of 'A' is treated with aq. NaOH, a liquid separates which contains N but not Cl. Treatment of the obtained liquid with nitrous acid followed by phenol gives orange precipitate. The compound 'A' is
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 48
Match List-I with List-II.
| List-I (Polymer) | List-II (Monomer) |
|---|---|
| A. Neoprene | I. Acrylonitrile |
| B. Teflon | II. Chloroprene |
| C. Acrilan | III. Tetrafluoroethene |
| D. Natural rubber | IV. Isoprene |
Choose the correct answer from the option given below:
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 49
Which of the following enhances the lathering property of soap?
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 50
Match List-I with List-II
| List-I | List-II |
|---|---|
| A. Glucose + HI | I. Gluconic acid |
| B. Glucose + $$Br_2$$ water | II. Glucose pentacetate |
| C. Glucose + acetic anhydride | III. Saccharic acid |
| D. Glucose + $$HNO_3$$ | IV. Hexane |
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 51
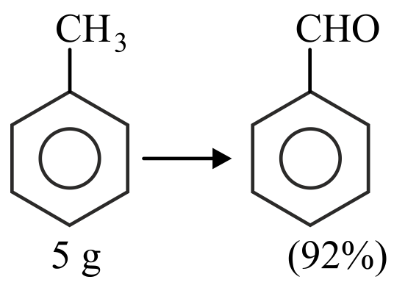
In the above reaction,5 g of toluene is converted into benzaldehyde with 92% yield. The amount of benzaldehyde produced is _____ $$\times 10^{-2}$$ g
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 52
The number of molecule(s) or ion(s) from the following having non-planar structure is ______.
$$NO_3^-$$, $$H_2O_2$$, $$BF_3$$, $$PCl_3$$, $$XeF_4$$, $$SF_4$$, $$XeO_3$$, $$PH_4^+$$, $$SO_3$$, $$[Al(OH)_4]^-$$
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 53
For a real gas at $$25^\circ C$$ temperature and high pressure (99 bar) the value of compressibility factor is 2, so the value of Van der Waal's constant 'b' should be _____ $$\times 10^{-2}$$ L mol$$^{-1}$$. (Given $$R = 0.083$$ L bar K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 54
A gas (Molar mass = 280 g mol$$^{-1}$$) was burnt in excess $$O_2$$ in a constant volume calorimeter and during combustion the temperature of calorimeter increased from 298.0 K to 298.45 K. If the heat capacity of calorimeter is 2.5 kJ K$$^{-1}$$ and enthalpy of combustion of gas is 9 kJ mol$$^{-1}$$ then amount of gas burnt is _____ g.
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 55
The normality of $$H_2SO_4$$ in the solution obtained on mixing 100 mL of 0.1 M $$H_2SO_4$$ with 50 mL of 0.1 M NaOH is _____ $$\times 10^{-1}$$ N.
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 56
When a certain amount of solid A is dissolved in 100 g of water at $$25^\circ C$$ to make a dilute solution, the vapour pressure of the solution is reduced to one-half of that of pure water. The vapour pressure of pure water is 23.76 mmHg. The number of moles of solute A added is
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 57
$$[A] \xrightarrow{} [B]$$
Reactant $$\to$$ Product
If formation of compound [B] follows the first order of kinetics and after 70 minutes the concentration of [A] was found to be half of its initial concentration. Then the rate constant of the reaction is $$x \times 10^{-6} s^{-1}$$. The value of $$x$$ is ____
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 58
Among the following ores Bauxite, Siderite, Cuprite, Calamine, Haematite, Kaolinite, Malachite, Magnetite, Sphalerite, Limonite, Cryolite, the number of principal ores of iron is
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 59
The oxidation state of manganese in the product obtained in a reaction of potassium permanganate and hydrogen peroxide in basic medium is
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Chemistry - Question 60
The spin only magnetic moment of the complex present in Fehling's reagent is _____ B.M. (Round off your answer to the nearest integer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




