Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The below P-V diagram represents the thermodynamic cycle of an engine, operating with an ideal mono-atomic gas. The amount of heat, extracted from the source in a single cycle, is:
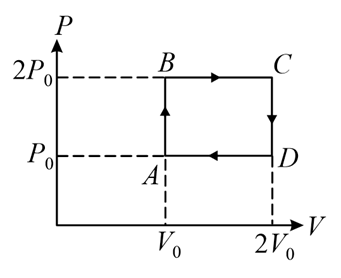
Heat is extracted from the source during the parts of the cycle where the heat change ($$Q$$) is positive. In the given P-V diagram, this occurs along the path $$A \to B \to C$$.
$$Q = \Delta U + W$$
$$\Delta U = n C_v \Delta T = \frac{3}{2} (P_f V_f - P_i V_i)$$ (for monoatomic gas)
Work Done ($$W_{ABC}$$):
Process $$A \to B$$ is isochoric, so $$W_{AB} = 0$$. Process $$B \to C$$ is isobaric expansion at $$2P_0$$. $$W_{ABC} = W_{BC} = P \Delta V = 2P_0(2V_0 - V_0) = 2P_0 V_0$$
Internal Energy Change ($$\Delta U_{ABC}$$):
From state $$A (P_0, V_0)$$ to state $$C (2P_0, 2V_0)$$,
$$\Delta U_{ABC} = \frac{3}{2} (P_C V_C - P_A V_A)$$
$$\Delta U_{ABC} = \frac{3}{2} (4P_0 V_0 - P_0 V_0) = \frac{3}{2} (3P_0 V_0) = \frac{9}{2} P_0 V_0$$
Total Heat Extracted ($$Q_{in}$$):
$$Q_{in} = \Delta U_{ABC} + W_{ABC}$$
$$Q_{in} = \left( \frac{9 + 4}{2} \right) P_0 V_0 = \frac{13}{2} P_0 V_0$$
Create a FREE account and get:
Educational materials for JEE preparation
