Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
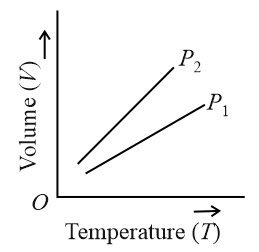
For a perfect gas, two pressures $$P_1$$ and $$P_2$$ are shown in figure. The graph shows
Concept:
For an ideal gas:
$$PV=nRT\ \Rightarrow\ \frac{V}{T}=\frac{nR}{P}\ $$
Analysis:
Slope of V-T graph:
slope=VT=nRP$$\text{slope}=\frac{V}{T}=\frac{nR}{P}$$
So, slope $$\propto\frac{1}{P}$$
From graph:
Line $$P_2$$ is steeper ⇒ larger slope ⇒ smaller pressure
$$\frac{nR}{P_2}>\frac{nR}{P_1}$$
$$\Rightarrow P_2$$
Final Answer:
$$P_2\ <\ P_1$$

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
