Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
A sample of an ideal gas is taken through the cyclic process $$ABCA$$ as shown in figure. It absorbs, 40 J of heat during the part $$AB$$, no heat during $$BC$$ and rejects 60 J of heat during $$CA$$. A work of 50 J is done on the gas during the part $$BC$$. The internal energy of the gas at $$A$$ is 1560 J. The work done by the gas during the part $$CA$$ is
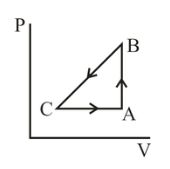
Use first law:
$$Q=\Delta U+W$$
(where W is work done by gas)
For process BC:
Given no heat exchange,
$$Q_{BC}=0$$
Work done on gas = 50 J
So work done by gas
$$W_{BC}=-50J$$
Thus
$$\Delta U_{BC}-50=0$$
$$ΔU_{BC}=50$$
So
$$U_C-U_B=50$$
For process AB:
$$Q_{AB}=40$$
From graph AB is vertical (constant volume), so
$$W_{AB}=0$$
Therefore
$$40=\Delta U_{AB}$$
$$U_B-U_A=40$$
Since
$$U_A=1560$$
$$U_B=1600$$
Then
$$U_C=1650$$
For process CA:
$$Q_{CA}=-60$$
Change in internal energy:
$$\Delta U_{CA}=U_A-U_C$$
$$=1560−1650=−90$$
Now
$$Q_{CA}=\Delta U_{CA}+W_{CA}\\-60=-90+W_{CA}$$
$$W_{CA}=30\text{ J}$$
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.