Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
In the reported figure, there is a cyclic process $$ABCDA$$ on a sample of 1 mol of a diatomic gas. The temperature of the gas during the process $$A \rightarrow B$$ and $$C \rightarrow D$$ are $$T_1$$ and $$T_2$$ ($$T_1 > T_2$$) respectively.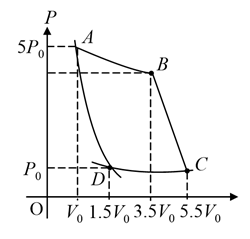
Choose the correct option out of the following for work done if processes $$BC$$ and $$DA$$ are adiabatic.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.