Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the following reactions sequence.
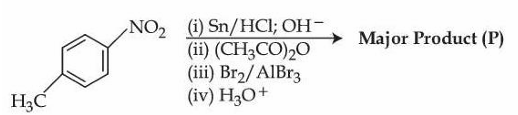
When the product (P) is subjected to Carius analysis using AgNO$$_3$$, 1.0 g of the product (P) will produce _____ g of the precipitate of AgBr. (Nearest Integer)
(Given: molar mass in g mol$$^{-1}$$ C: 12, H: 1, O: 16, N: 14, Br: 80, Ag: 108)
Correct Answer: 1
Step 1 : Reduction of the nitro group
$$4\text{-methyl-}NO_2\text{-benzene}
\xrightarrow[\;OH^-]{Sn/HCl} 4\text{-methyl aniline }(p\text{-toluidine})$$
Step 2 : Acetylation of the amino group (protection)
$$p\text{-toluidine}\xrightarrow{(CH_3CO)_2O}
p\text{-methyl acetanilide }( -NHCOCH_3 )$$
Step 3 : Electrophilic bromination
In acetanilide the $$-NHCOCH_3$$ group is ortho-/para-directing.
The para position is already occupied by $$-CH_3$$, so bromine enters one of the two ortho positions; steric factors give the single major product.
Major brominated product after step 3:
$$\text{2-bromo-4-methyl acetanilide}$$
Step 4 : Hydrolytic deprotection
$$\text{2-bromo-4-methyl acetanilide} \xrightarrow{H_3O^+}
\text{2-bromo-4-methyl aniline}$$
Thus the final compound (P) is 2-bromo-4-methyl aniline.
Step 5 : Molecular formula and molar mass of (P)
Ring carbons: 6, extra methyl carbon: 1 ⇒ $$C_7$$
Ring hydrogens: 3 (after three substitutions) + methyl H: 3 + amino H: 2 ⇒ $$H_8$$
One nitrogen and one bromine ⇒ $$N_1Br_1$$
Molecular formula: $$C_7H_8BrN$$
Molar mass (g mol$$^{-1}$$): $$7(12) + 8(1) + 80 + 14 = 84 + 8 + 80 + 14 = 186$$
Step 6 : Carius (halogen) analysis
Each mole of compound has one Br atom, giving one mole of $$AgBr$$.
Molar mass of $$AgBr = 108 + 80 = 188\text{ g mol}^{-1}$$
Moles of (P) in 1.0 g sample:
$$n = \frac{1.0}{186} = 5.38\times10^{-3}\;\text{mol}$$
Mass of $$AgBr$$ formed:
$$m = n \times 188 = (5.38\times10^{-3})\times188 \approx 1.01\text{ g}$$
Nearest integer = 1 g.
Final Answer: 1
Create a FREE account and get:
Educational materials for JEE preparation
