Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
For a first order reaction A $$\to$$ B
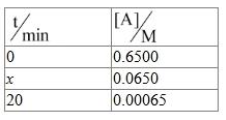
$$x$$ = _________ min. (Nearest integer)
Correct Answer: 7
For a first-order reaction the integrated rate law is
$$\ln\!\left(\frac{[A]_0}{[A]}\right)=kt$$
Here $$[A]_0=0.6500\text{ M}$$ at $$t=0$$.
We first evaluate the rate constant $$k$$ using the data at $$t=20\ \text{min}$$, $$[A]=0.00065\text{ M}$$.
$$\ln\!\left(\frac{0.6500}{0.00065}\right)=k(20)$$
Since $$\dfrac{0.6500}{0.00065}=1000$$, and $$\ln(1000)=\ln(10^3)=3\ln10=3(2.302585)=6.907755$$, we get
$$k=\frac{6.907755}{20}=0.3454\ \text{min}^{-1}$$
Next, let $$x$$ be the time (in minutes) when $$[A]=0.0650\text{ M}$$.
Applying the same rate law:
$$\ln\!\left(\frac{0.6500}{0.0650}\right)=k\,x$$
The ratio is $$\dfrac{0.6500}{0.0650}=10$$, hence $$\ln10=2.302585$$.
Therefore
$$x=\frac{\ln10}{k}=\frac{2.302585}{0.3454}=6.67\ \text{min}$$
Rounding to the nearest whole number gives $$x\approx7\ \text{minutes}$$.
Final Answer: 7
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
