Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
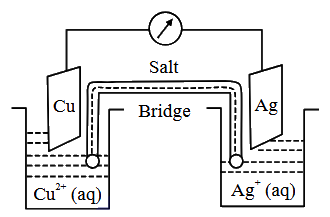
1 Faraday electricity was passed through $$Cu^{2+}$$ (1.5 M, 1 L)/Cu and 0.1 Faraday was passed through $$Ag^+$$ (0.2 M, 1 L)/Ag electrolytic cells. After this the two cells were connected as shown below to make an electrochemical cell. The emf of the cell thus formed at 298 K is ______. (Given: $$E^0_{Cu^{2+}/Cu} = 0.34$$ V, $$E^0_{Ag^+/Ag} = 0.8$$ V, $$\frac{2.303RT}{F} = 0.06$$ V)
Correct Answer: 400
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.