Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
For the thermal decomposition of $$N_{2}O_{5}(g)$$ at constant volume, the following table can be formed, for the reaction mentioned below. $$2 N_{2}O_{5}(g)\rightarrow 2 N_{2}O_{4}(g)+O_{2}(g)$$
$$x= .... \times 10^{-3}$$ atm [nearest integer] Given : Rate constant for the reaction is $$4.606 \times 10^{-2} s^{-1}.$$
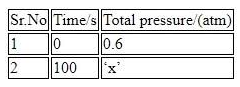
Correct Answer: 897

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
