Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the following sequence of reactions to give the major product $$(X)$$:
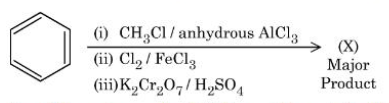
$$P$$ g of the major product $$(X)$$ formed is reacted with $$\text{NaHCO}_3$$ solution to liberate a gas which occupied 11.2 dm³ at STP. $$P$$ = _____ g.
( Given molar mass in g $$mol^{-1} H:1,C:12,O:16,Cl:35.5$$)
Correct Answer: 78
Create a FREE account and get:
Educational materials for JEE preparation
