Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
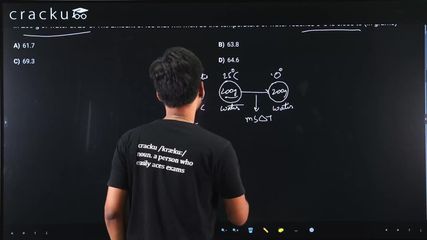
The specific heat of water = 4200 J kg$$^{-1}$$ K$$^{-1}$$ and the latent heat of ice = $$3.4 \times 10^5$$ J kg$$^{-1}$$. 100 grams of ice at 0°C is placed in 200 g of water at 25°C. The amount of ice that will melt as the temperature of water reaches 0°C is close to (in grams)
We start by noting that the only significant energy exchange is between the warm water and the ice; the container and surroundings are assumed perfectly insulating. Because the ice is already at its melting point (0 °C) and the water finally cools to 0 °C, no part of the ice-water mixture ever rises above 0 °C. Hence the warm water will simply give up some of its thermal energy, cooling from 25 °C down to 0 °C, and that energy will go exclusively into melting a certain mass of ice.
First we calculate the heat lost by the water as it cools. The specific heat capacity formula is stated as
$$Q = m\,c\,\Delta T,$$
where $$m$$ is the mass of the substance, $$c$$ its specific heat capacity, and $$\Delta T$$ the change in temperature.
The warm water has
$$m_w = 200 \text{ g} = 0.200 \text{ kg},$$
$$c_w = 4200 \text{ J\,kg}^{-1}\text{K}^{-1},$$
$$\Delta T_w = 25^\circ\text{C} - 0^\circ\text{C} = 25 \text{ K}.$$
Substituting these values, we obtain
$$Q_{\text{lost}} = (0.200)\,(4200)\,(25) \text{ J}.$$
Now we multiply step by step:
$$4200 \times 25 = 105,000,$$
$$0.200 \times 105,000 = 21,000.$$
So,
$$Q_{\text{lost}} = 21,000 \text{ J}.$$
This 21,000 J of energy is absorbed by a portion of the ice, melting it while itself staying at 0 °C. For melting, the relevant formula is the latent heat relation
$$Q = m L,$$
where $$m$$ is the mass melted and $$L$$ is the latent heat of fusion of ice.
Given
$$L = 3.4 \times 10^{5} \text{ J\,kg}^{-1},$$
we set
$$Q_{\text{lost}} = Q_{\text{gained}},$$
$$21,000 = m\,L.$$
Solving for $$m$$ we have
$$m = \frac{21,000}{3.4 \times 10^{5}} \text{ kg}.$$
Carrying out the division step by step, we write
$$\frac{21,000}{340,000} = 0.0617647\ldots \text{ kg}.$$
To convert this to grams we multiply by 1000:
$$0.0617647\ldots \text{ kg} \times 1000 = 61.7647\ldots \text{ g}.$$
Rounded sensibly to three significant figures, the mass of ice that melts is approximately
$$\boxed{61.7 \text{ g}}.$$
Hence, the correct answer is Option A.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
