Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Match the LIST-I with LIST-II
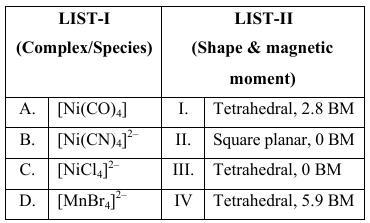
Choose the correct answer from the options given below :
First write the ground-state electronic configurations of the central metal atoms.
Ni : $$[Ar]\,3d^{8}\,4s^{2}$$
Mn : $$[Ar]\,3d^{5}\,4s^{2}$$
Next, find the oxidation state of the metal in each complex.
$$[Ni(CO)_4]$$ : CO is neutral ⇒ Ni is $$0$$-oxidation.
$$[Ni(CN)_4]^{2-}$$ : $$4(\!-1)$$ from $$CN^-$$ gives $$\;x-4=-2\Rightarrow x=+2\;(Ni^{2+})$$.
$$[NiCl_4]^{2-}$$ : $$4(\!-1)$$ from $$Cl^-$$ gives $$\;x-4=-2\Rightarrow x=+2\;(Ni^{2+})$$.
$$[MnBr_4]^{2-}$$ : $$4(\!-1)$$ from $$Br^-$$ gives $$\;x-4=-2\Rightarrow x=+2\;(Mn^{2+})$$.
Now list the $$d$$-electron counts.
Ni(0) $$3d^{10}$$ (after promotion-hybridisation will pair all 10).
Ni(II) $$3d^{8}$$.
Mn(II) $$3d^{5}$$.
Identify each ligand as strong- or weak-field.
CO and $$CN^-$$ are strong-field (cause pairing).
$$Cl^-$$ and $$Br^-$$ are weak-field (do not cause pairing).
Determine hybridisation, geometry and unpaired electrons one by one.
Case A: $$[Ni(CO)_4]$$
CO is strong field; Ni is in $$0$$ state. The 3d orbitals are fully paired and do not participate in hybridisation. The complex uses $$sp^3$$ hybridisation (4s + 3p) ⇒ tetrahedral. With all electrons paired, $$n=0$$ unpaired ⇒ $$\mu =0\;BM$$. Hence: Tetrahedral, 0 BM (Choice III).
Case B: $$[Ni(CN)_4]^{2-}$$
Ni$$^{2+}$$ is $$3d^{8}$$. $$CN^-$$ is strong field, so the electrons pair up giving configuration $$t_{2g}^{6}e_g^{2}$$ with no unpaired electrons. Square-planar geometry arises from $$dsp^2$$ hybridisation. $$n=0$$ ⇒ $$\mu=0\;BM$$. Hence: Square planar, 0 BM (Choice II).
Case C: $$[NiCl_4]^{2-}$$
Ni$$^{2+}$$ is $$3d^{8}$$ and $$Cl^-$$ is weak field, so no additional pairing occurs. The complex adopts $$sp^3$$ hybridisation ⇒ tetrahedral. For a tetrahedral high-spin $$d^{8}$$ ion there are $$n=2$$ unpaired electrons, so
$$\mu=\sqrt{n(n+2)}=\sqrt{2\times4}\approx2.8\;BM$$. Hence: Tetrahedral, 2.8 BM (Choice I).
Case D: $$[MnBr_4]^{2-}$$
Mn$$^{2+}$$ is $$3d^{5}$$, and the weak-field $$Br^-$$ keeps it high-spin. With $$sp^3$$ hybridisation the geometry is tetrahedral. All five $$d$$ electrons remain unpaired, $$n=5$$, so
$$\mu=\sqrt{5(5+2)}=\sqrt{35}\approx5.9\;BM$$. Hence: Tetrahedral, 5.9 BM (Choice IV).
Collecting the results:
A → III B → II C → I D → IV
Comparing with the given options, this matches Option C.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
