Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Given below are two statements:
Statement (I) : for

, all three possible structures may be drawn.
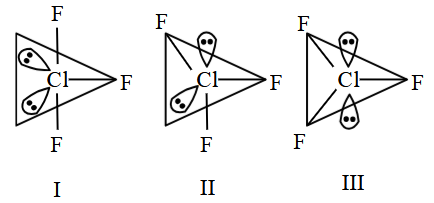
Statement (II) : Structure III is most stable, as the orbitals having the lone pairs are axial, where the $$lp-bp$$ repulsion is minimum.
In the light of the above statements, choose the most appropriate answer from the options given below:
The central atom $$Cl$$ in $$ClF_3$$ has $$7$$ valence electrons. Three of them are used to form $$\sigma$$-bonds with three $$F$$ atoms and the remaining $$4$$ electrons constitute two lone pairs. Hence the steric number is $$5$$ ( $$= 3\; \text{bond pairs} + 2\; \text{lone pairs}$$ ).
For steric number $$5$$, the electron-pair geometry is $$\text{trigonal bipyramidal}$$. The five positions are
• two axial (directly opposite, $$180^{\circ}$$ apart)
• three equatorial (coplanar, $$120^{\circ}$$ apart)
Placing two lone pairs and three bond pairs in these five positions can, in principle, give the following three arrangements:
Case I: both lone pairs axial $$\rightarrow$$ all three $$F$$ atoms equatorial.
Case II: one lone pair axial, one lone pair equatorial $$\rightarrow$$ two $$F$$ atoms equatorial, one axial.
Case III: both lone pairs equatorial $$\rightarrow$$ one $$F$$ atom equatorial, two $$F$$ atoms axial (T-shaped).
Thus, as statement (I) says, all three structures can indeed be drawn; so Statement I is correct.
According to VSEPR theory the order of repulsions is $$lp\!-\!lp \; \gt \; lp\!-\!bp \; \gt \; bp\!-\!bp$$. An axial position is at $$90^{\circ}$$ to three other positions, whereas an equatorial position is at $$90^{\circ}$$ to only two. Therefore a lone pair experiences less repulsion when it occupies an equatorial site.
Hence Case III, in which both lone pairs occupy the equatorial positions, has the minimum total repulsion and is the most stable. Statement (II) claims the most stable structure has lone pairs in axial orbitals, which is opposite to the VSEPR prediction. Therefore Statement II is incorrect.
So, Statement I is correct while Statement II is incorrect. Hence the correct option is Option B.
Create a FREE account and get:
Educational materials for JEE preparation
