Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Match the LIST-I with LIST-II.
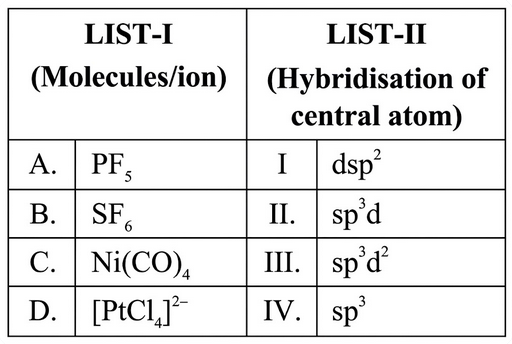
Choose the correct answer from the options given below :
The hybridisation of a central atom is obtained from its steric number (total number of $$\sigma$$-bonds + lone pairs).
For transition-metal complexes we also look at the observed geometry.
Case A: $$PF_5$$
Phosphorus forms five $$\sigma$$-bonds with fluorine and has no lone pair.
Hence steric number $$=5$$ $$\Rightarrow$$ hybridisation $$= sp^3d$$ (trigonal bipyramidal).
Therefore $$PF_5$$ matches List-II entry II.
Case B: $$SF_6$$
Sulphur forms six $$\sigma$$-bonds with fluorine and has no lone pair.
Steric number $$=6$$ $$\Rightarrow$$ hybridisation $$= sp^3d^2$$ (octahedral).
Therefore $$SF_6$$ matches List-II entry III.
Case C: $$Ni(CO)_4$$
Nickel in $$Ni(CO)_4$$ is in the zero oxidation state and the complex is tetrahedral.
A tetrahedral arrangement uses four equivalent hybrid orbitals: $$sp^3$$.
Therefore $$Ni(CO)_4$$ matches List-II entry IV.
Case D: $$[PtCl_4]^{2-}$$
The ion is square-planar; Pt(II) uses one $$d$$, one $$s$$ and two $$p$$ orbitals forming $$dsp^2$$ hybrids.
Therefore $$[PtCl_4]^{2-}$$ matches List-II entry I.
Collecting all results:
A → II, B → III, C → IV, D → I.
The correct option is Option A.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.