Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
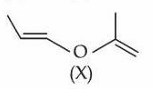
A molecule (X) with the following structure under mild acidic condition is hydrolysed to produce (Y) and (Z). Identify the correct statements about (Y) and (Z).
A. Both (Y) and (Z) have same molar mass.
B. (Y) and (Z) can be distinguished from each other by NaHCO$$_3$$.
C. (Y) and (Z) react with HCN with same rates.
D. (Y) and (Z) undergo addition reaction with 2,4-DNP.
Choose the correct answer from the options given below :
The given compound $$(X)$$ is an acetal.
Under mild acidic conditions, acetals undergo hydrolysis to give back the two carbonyl compounds from which they were originally obtained.
On hydrolysis, $$(X)$$ breaks at the two $$C-O$$ bonds adjacent to the acetal carbon and furnishes the following products:
$$\displaystyle (X)\; \xrightarrow[\text{H}_2O]{\text{dil.\,H}^+}\; (Y)\;+\; (Z)$$
The two fragments obtained are
$$\text{propanal :}\; CH_3CH_2CHO \; (Y)$$
$$\text{acetone :}\; (CH_3)_2CO \; (Z)$$
Both have the same molecular formula $$C_3H_6O$$, so their molar mass is identical:
$$M_r(CH_3CH_2CHO)=58\;{\rm g\,mol^{-1}}$$
$$M_r((CH_3)_2CO)=58\;{\rm g\,mol^{-1}}$$
Let us now analyse each statement:
Statement A: “Both (Y) and (Z) have same molar mass.”
As shown above, this is correct.
Statement B: “(Y) and (Z) can be distinguished from each other by NaHCO$$$_3$$.”
$$NaHCO_3$$ gives effervescence only with acids that are stronger than $$H_2CO_3$$ (mainly carboxylic acids and sulfonic acids). Aldehydes and ketones do not react. Hence neither propanal nor acetone shows any reaction; this test cannot distinguish them. Statement B is false.
Statement C: “(Y) and (Z) react with HCN with same rates.”
The rate of cyanohydrin formation depends on both the electronic nature of the carbonyl carbon and steric hindrance. Aldehydes (propanal) are less hindered and more polarised than ketones (acetone); therefore propanal reacts faster with $$HCN$$ than acetone. Rates are not the same, making Statement C false.
Statement D: “(Y) and (Z) undergo addition reaction with 2,4-DNP.”
2,4-Dinitrophenylhydrazine adds to any aldehyde or ketone to form a yellow/orange hydrazone precipitate. Both propanal and acetone give a positive 2,4-DNP test. Statement D is correct.
Thus only Statements A and D are correct.
Option D $$\longrightarrow\ $$ A and D Only
Create a FREE account and get:
Educational materials for JEE preparation
