Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
When sec-butylcyclohexane reacts with bromine in the presence of sunlight, the major product is :
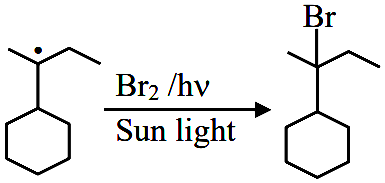
In the free radical bromination of sec-butylcyclohexane, bromine selectively attacks the most stable radical intermediate, which is the tertiary ($$3^\circ$$) carbon at the ring junction (Carbon I). This specific position is prioritized over the side-chain tertiary carbon because it is stabilized by a higher number of $$\alpha$$-hydrogen atoms through hyperconjugation.

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
