Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The descending order of acidity among the following compounds is:
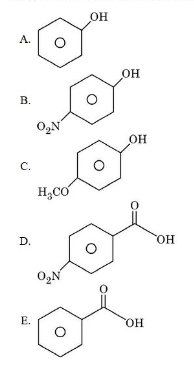
choose the correct answer from the options given below:
Acid strength is decided by the stability of the conjugate base that remains after loss of $$H^+$$. A more stable conjugate base means a stronger acid.
The stabilising factors to look for are: (i) resonance delocalisation, (ii) -I (inductive) electron withdrawal, (iii) +M (mesomeric) electron donation which has the opposite effect.
Step 1 : Separate carboxylic acids from phenols/alcohols
Carboxylic acids are far more acidic (pKₐ ≈ 4-5) than phenols (pKₐ ≈ 10) and aliphatic alcohols (pKₐ ≈ 16).
Hence any carboxylic acid in the list will automatically be the strongest.
The two carboxylic acids are:
(D) p-nitrobenzoic acid and (E) benzoic acid.
Step 2 : Order the two carboxylic acids
The conjugate base of p-nitrobenzoic acid bears a $$\mathrm{NO_2}$$ group at the para position. $$\mathrm{NO_2}$$ is a strong -M and -I group that withdraws electron density, dispersing the negative charge by resonance as well as inductively.
Therefore it stabilises the benzoate ion much more than does a hydrogen (in plain benzoic acid).
So $$\text{acidity: } (D) \gt (E).$$
Step 3 : Compare phenolic compounds
Among phenols acidity depends on the substituent on the ring.
(B) o-Nitrophenol : $$\mathrm{NO_2}$$ is −M and −I, stabilising the phenoxide ion by delocalising the charge. However, intramolecular H-bonding in o-nitrophenol slightly reduces its ability to form stronger hydrogen bonds with solvent, yet overall it is still more acidic than unsubstituted phenol.
(C) p-Methoxyphenol : $$\mathrm{OCH_3}$$ shows a strong +M (electron-donating) effect which pushes electron density toward the oxygen, destabilising the phenoxide ion. Thus its acidity is lower than that of phenol itself.
Hence $$\text{acidity: } (B) \gt (C).$$
Step 4 : Insert the alcohol
(A) Cyclohexanol possesses no resonance stabilisation of its alkoxide ion; only a weak −I effect of the ring is present.
Consequently, cyclohexanol is far less acidic than any phenol but a little more acidic than p-methoxyphenol because the +M donation in (C) actually destabilises that phenoxide even more than the absence of resonance in an aliphatic alcohol.
Combining every block:
(D) p-nitrobenzoic acid > (E) benzoic acid > (B) o-nitrophenol > (A) cyclohexanol > (C) p-methoxyphenol.
Therefore the required descending order of acidity is
$$D \gt E \gt B \gt A \gt C$$
Option D which is: D > E > B > A > C
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
