Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Given below are two statements :
Statement I : Due to increase in van der Waals forces, the order of boiling points is CH$$_3$$CH$$_2$$CH$$_2$$I > CH$$_3$$CH$$_2$$I > CH$$_3$$I.
Statement II : As
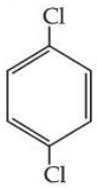
is more symmetric, its melting point is higher than
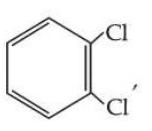
, however its boiling point is lower than
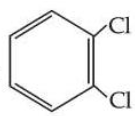
.
In the light of the above statements, choose the correct answer from the options given below :
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
