Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the following two reactions A and B.
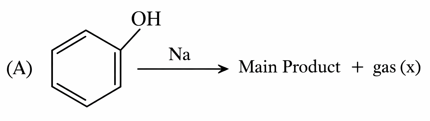
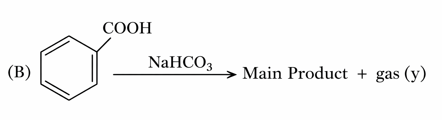
Numerical value of [ molar mass of x + molar mass of y] is __
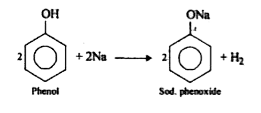
gas(x) = $$H_2$$
molar mass (x) = 2g/mol
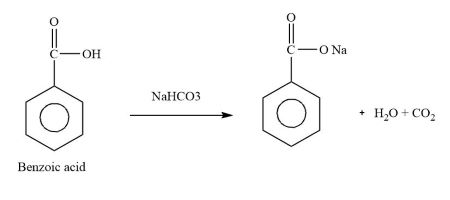
gas(y) = $$CO_2$$
molar mass (y) = 44 g/mol
molar mass of (x) + molar mass of (y): 2 + 44 = 46g/mol

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
