Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
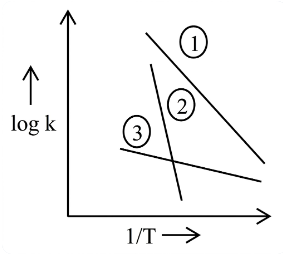
Consider the following plots of log of rate constant k (log k) vs $$\frac{1}{T}$$ for three different reactions. The correct order of activation energies of these reactions is :
Create a FREE account and get:
Educational materials for JEE preparation
