Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
An alkene $$(X)$$ on ozonolysis followed by reduction gives following products:
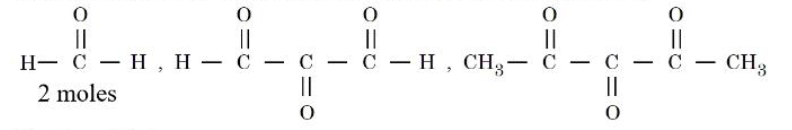
The alkene $$(X)$$ is :
During reductive ozonolysis each carbon of the $$C=C$$ bond is cleaved to give a carbonyl group. Hence, if the two products are acetone (propanone) $$\big(CH_3COCH_3\big)$$ and acetaldehyde (ethanal) $$\big(CH_3CHO\big)$$, then the carbon atoms that carry the carbonyl oxygens in these two molecules were the two carbon atoms of the original double bond.
To reconstruct the parent alkene we therefore
1. remove the O atoms from the two carbonyl groups, and
2. join the two carbon atoms by a double bond.
Doing this gives
$$(CH_3)_2C=CHCH_3$$
This molecule is named 2-methyl-2-butene.
Verification: On cleaving the $$C_2=C_3$$ bond of 2-methyl-2-butene by ozonolysis, carbon-2 (which bears two $$CH_3$$ groups) becomes acetone, while carbon-3 (bearing one $$CH_3$$ and one H) becomes acetaldehyde, exactly matching the given products.
Hence the required alkene $$X$$ is 2-methyl-2-butene, which is listed as Option B.
Create a FREE account and get:
Educational materials for JEE preparation
